This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1053
From Proteopedia
| Line 30: | Line 30: | ||
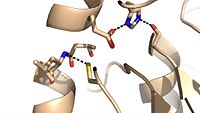
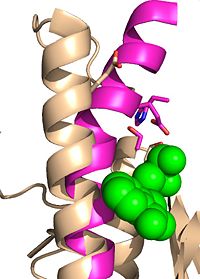
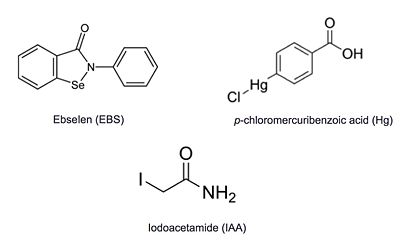
| - | Ag85C can be inhibited by [http://en.wikipedia.org/wiki/Ebselen ebselen] covalently bound to the sulfur of C209. Ebselen is a thiol-modifying agent that serves as an electrophile for the C209 that results in a sulfur-selenium bond. Co-crystallization of ebselen with Ag85C provides an explanation for the mechanism of ebselen-based inhibition. The addition of ebselen increases the distance between C209 and L232-T31, which effectively disrupts the interaction that holds the α9 helix in the active conformation. Furthermore, the bulk of ebselen creates steric hindrance with the α9 helix residues | + | Ag85C can be inhibited by [http://en.wikipedia.org/wiki/Ebselen ebselen] covalently bound to the sulfur of C209. Ebselen is a thiol-modifying agent that serves as an electrophile for the C209 that results in a sulfur-selenium bond. Co-crystallization of ebselen with Ag85C provides an explanation for the mechanism of ebselen-based inhibition. The addition of ebselen increases the distance between C209 and L232-T31, which effectively disrupts the interaction that holds the α9 helix in the active conformation. Furthermore, the bulk of ebselen creates steric hindrance with the α9 helix residues, which can be seen in the figure to the right. Relaxation of the α9 helix removes E228 and H260, which now interacts with S148, from the active site. The absence of these residues decreases the nucleophilicity of the S124 alcohol which decreases serine hydrolytic activity. |
Relaxed alpha helix scene <scene name='69/694220/Inhibited_relaxed_helix/1'>relaxed</scene> | Relaxed alpha helix scene <scene name='69/694220/Inhibited_relaxed_helix/1'>relaxed</scene> | ||
Revision as of 18:51, 27 April 2015
| This Sandbox is Reserved from 02/09/2015, through 05/31/2016 for use in the course "CH462: Biochemistry 2" taught by Geoffrey C. Hoops at the Butler University. This reservation includes Sandbox Reserved 1051 through Sandbox Reserved 1080. |
To get started:
More help: Help:Editing |
Background
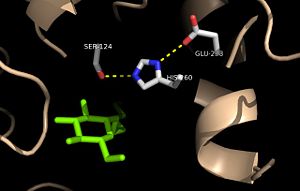
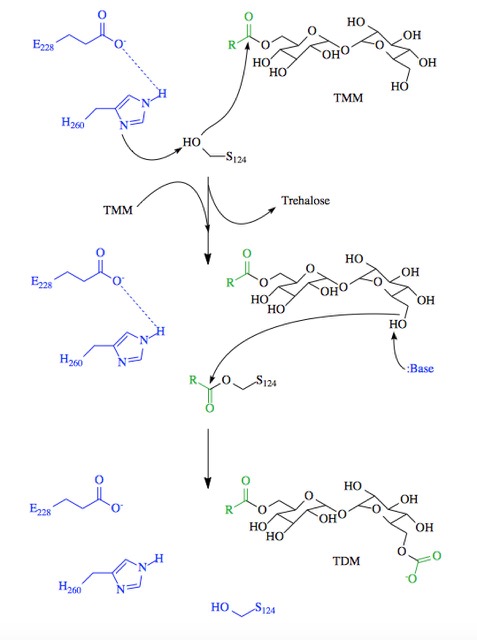
The antigen 85 (ag85) complex in Mycobacterium tuberculosis, which is responsible for causing the disease Tuberculosis, is composed of three intracellular membrane proteins: Ag85A, B, and C. The Ag85 complex is a major component of the cell wall, with each protein catalyzing the transfer of important cell wall constituents into the membrane. [1] The cell wall of Mycobacterium tuberculosis is composed of three primary molecules: peptidoglycans, arbinogalactans, and mycolic acids. Ag85C is of particular interest due to its transfer of mycolic acids, which is one of the major components in determining cell wall integrity. The mycolic acids are responsible for forming the outermost layer of the cell wall. Mycolic acids have a long fatty acid chain and exhibit extreme hydrophobicity, which effectively creates a hydrophobic envelope surrounding the bacterium. The hydrophobic envelope created by they mycolic acids creates a barrier against small hydrophilic molecules, such as Tuberculosis antibiotics. By targeting this mycoloyltransferase activity, inhibition of Ag85C offers potential for cell wall disruption and subsequent antibiotic targeting for normally drug-resistant Mycotaberia tuberculosis. [2]
Structure
| |||||||||||
References
- ↑ Ronning DR, Klabunde T, Besra GS, Vissa VD, Belisle JT, Sacchettini JC. Crystal structure of the secreted form of antigen 85C reveals potential targets for mycobacterial drugs and vaccines. Nat Struct Biol. 2000 Feb;7(2):141-6. PMID:10655617 doi:10.1038/72413
- ↑ Jackson M, Raynaud C, Laneelle MA, Guilhot C, Laurent-Winter C, Ensergueix D, Gicquel B, Daffe M. Inactivation of the antigen 85C gene profoundly affects the mycolate content and alters the permeability of the Mycobacterium tuberculosis cell envelope. Mol Microbiol. 1999 Mar;31(5):1573-87. PMID:10200974
- ↑ Favrot L, Lajiness DH, Ronning DR. Inactivation of the Mycobacterium tuberculosis Antigen 85 complex by covalent, allosteric inhibitors. J Biol Chem. 2014 Jul 14. pii: jbc.M114.582445. PMID:25028518 doi:http://dx.doi.org/10.1074/jbc.M114.582445
- ↑ Ronning DR, Klabunde T, Besra GS, Vissa VD, Belisle JT, Sacchettini JC. Crystal structure of the secreted form of antigen 85C reveals potential targets for mycobacterial drugs and vaccines. Nat Struct Biol. 2000 Feb;7(2):141-6. PMID:10655617 doi:10.1038/72413