This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Connexin
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
==Connexin structure== | ==Connexin structure== | ||
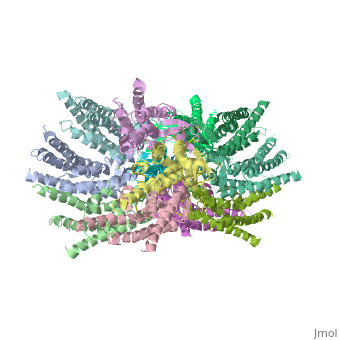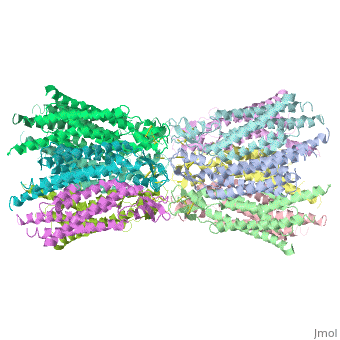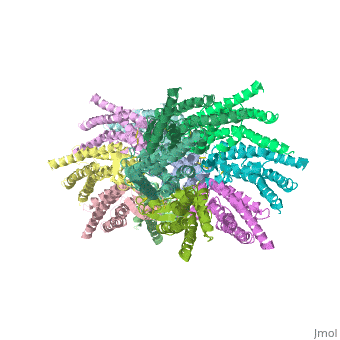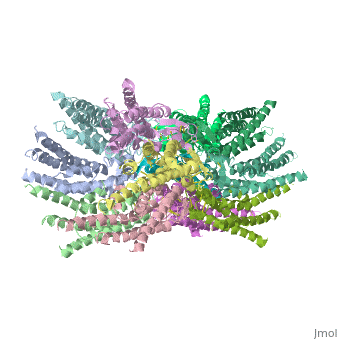
Connexins are integral α-hellical transmembrane proteins that form intercellular channels in vertebrates <scene name='70/701426/Connexons_secondary_structure/1'>connexons secondary structure</scene>. Six connexins form a hexamerical assembly, known as connexon or hemichannel <scene name='70/701426/Connexin_26_basic_structure/2'>Connexon as a six connexins assembly</scene>, which delineates an aqueous pore with a minimum diameter of ∼1.2 nm. When two hemichannels from adjacent cells dock and join, leaving a gap of ∼2–3 nm, they may form an intercellular [http://www.uniprot.org/uniprot/P29033 gap junction channel] which spans the two pαlasma membranes and allows the exchange of cytoplasmic molecules with size up to ∼1 kDa. | Connexins are integral α-hellical transmembrane proteins that form intercellular channels in vertebrates <scene name='70/701426/Connexons_secondary_structure/1'>connexons secondary structure</scene>. Six connexins form a hexamerical assembly, known as connexon or hemichannel <scene name='70/701426/Connexin_26_basic_structure/2'>Connexon as a six connexins assembly</scene>, which delineates an aqueous pore with a minimum diameter of ∼1.2 nm. When two hemichannels from adjacent cells dock and join, leaving a gap of ∼2–3 nm, they may form an intercellular [http://www.uniprot.org/uniprot/P29033 gap junction channel] which spans the two pαlasma membranes and allows the exchange of cytoplasmic molecules with size up to ∼1 kDa. | ||
| - | The height of the modelled structure of the gap junction channel without disordered cytoplasmic loop and C-terminal segment is approximately | + | The height of the modelled structure of the gap junction channel without disordered cytoplasmic loop and C-terminal segment is approximately 155Å. The transmembrane region and membrane surfaces were deduced from the distribution of hydrophobic and aromatic amino acid residues along the noncrystallographic six-fold axis It is a tsuzumi shape, a traditional Japanese drum.<ref name='Structure'/> [[Image:distances a.jpg]] |
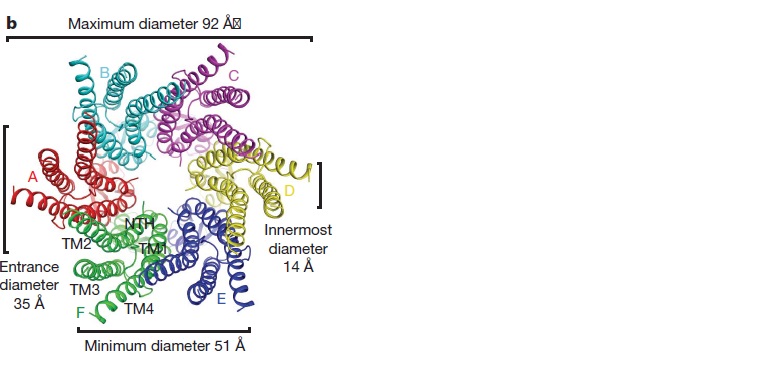
| - | The protomers in each hexameric connexon are related by a sixfold non-crystallographic symmetry (NCS) axis perpendicular to the membrane plane . The transmembrane region of the channel is | + | The protomers in each hexameric connexon are related by a sixfold non-crystallographic symmetry (NCS) axis perpendicular to the membrane plane . The transmembrane region of the channel is 38Å thick.TM2 extends about 19Å from the membrane surface into the cytoplasm. The extracellular region of the connexon extends 23Å from the membrane surface and interdigitates to the opposite connexon by 6Å, resulting in the intercellular ‘gap’ of 40Å. The extracellular lobes are not protruding so much, as indicated by the structural analyses of split gap junction channels with atomic force microscopy and electron microscopy. The relatively flat lobes could be attributed to the conformational change of the extracellular region induced by the docking of two connexons. The diameter of the connexon is biggest at the cytoplasmic side |
| - | of the membrane, | + | of the membrane, 92Å , and smallest at the extracellular side, 51Å . |
| - | Viewed from the top, the channel looks like a ‘hexagonal nut’ with a pore in the centre .The diameter of the pore is about | + | Viewed from the top, the channel looks like a ‘hexagonal nut’ with a pore in the centre .The diameter of the pore is about 40Å at the cytoplasmic side of the channel, narrowing to 14Å near the extracellular membrane surface and then widening to 25Å in the extracellular space.<ref name='Structure'/> |
[[Image:distances b.jpg]] | [[Image:distances b.jpg]] | ||
==Structure of the cx26 protomer:== | ==Structure of the cx26 protomer:== | ||
Revision as of 10:12, 17 May 2015
| |||||||||||
References
- ↑ Zonta F, Buratto D, Cassini C, Bortolozzi M, Mammano F. Molecular dynamics simulations highlight structural and functional alterations in deafness-related M34T mutation of connexin 26. Front Physiol. 2014 Mar 4;5:85. doi: 10.3389/fphys.2014.00085. eCollection 2014. PMID:24624091 doi:http://dx.doi.org/10.3389/fphys.2014.00085
- ↑ 2.0 2.1 2.2 2.3 2.4 Suga M, Maeda S, Nakagawa S, Yamashita E, Tsukihara T. A description of the structural determination procedures of a gap junction channel at 3.5 A resolution. Acta Crystallogr D Biol Crystallogr. 2009 Aug;65(Pt 8):758-66. Epub 2009, Jul 10. PMID:19622859 doi:http://dx.doi.org/10.1107/S0907444909014711
- ↑ 3.0 3.1 Ambrosi C, Walker AE, Depriest AD, Cone AC, Lu C, Badger J, Skerrett IM, Sosinsky GE. Analysis of trafficking, stability and function of human connexin 26 gap junction channels with deafness-causing mutations in the fourth transmembrane helix. PLoS One. 2013 Aug 15;8(8):e70916. doi: 10.1371/journal.pone.0070916. eCollection, 2013. PMID:23967136 doi:http://dx.doi.org/10.1371/journal.pone.0070916
- ↑ 4.0 4.1 Oshima A, Tani K, Toloue MM, Hiroaki Y, Smock A, Inukai S, Cone A, Nicholson BJ, Sosinsky GE, Fujiyoshi Y. Asymmetric Configurations and N-terminal Rearrangements in Connexin26 Gap Junction Channels. J Mol Biol. 2011 Jan 21;405(3):724-35. Epub 2010 Nov 20. PMID:21094651 doi:10.1016/j.jmb.2010.10.032
Proteopedia Page Contributors and Editors (what is this?)
Safaa Salah Hussiesy, Michal Harel, Doaa Naffaa, Jaime Prilusky