This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
PLC beta 3 Gq
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
== Introduction == | == Introduction == | ||
| - | Phospholipase C (PLC) catalyzes the hydrolysis of phosphatidylinositol 4,5-bisphosphate [ | + | Phospholipase C beta 3 (PLC-β3) catalyzes the hydrolysis of phosphatidylinositol 4,5-bisphosphate [PIP2] to the second messengers inositol 1,4,5-trisphosphate [IP3] and diacylglycerol [DAG] in an essential step for many physiological cascades. When the receptor is stimulated by ligand of some kind it increase exchange of guanosine diphosphate (GDP) for guanosine triphosphate (GTP) on Gαq. GTP-bound Gαq activates PLC- β3, and PLC- β3 increases up to three orders of magnitude the rate of hydrolysis of GTP by its activating G protein. This is a unique mechanism in which the PLC-β3 enzyme has the ability to terminate the Gαq protein signal in addition to being activated by it.<ref>PMID:20966218</ref> <ref>PMID:23880553</ref> |
== Structural highlights == | == Structural highlights == | ||
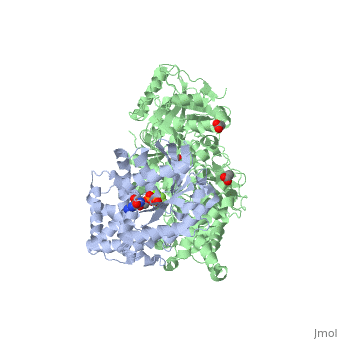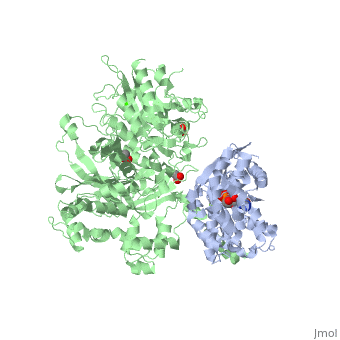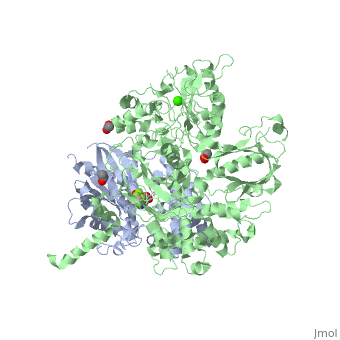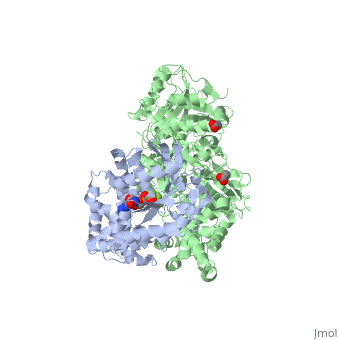
| - | The Gαq subunit consists two domains, one is the GTPase domain and the other is the alpha helical domain. These domains include three regions called <scene name='70/701452/Fig2/ | + | The Gαq subunit consists two domains, one is the GTPase domain and the other is the alpha helical domain. These domains include three regions called <scene name='70/701452/Fig2/5'>switch regions I-III</scene>. |
<scene name='70/701452/Fig1/9'>The PLC- β3 has several domains</scene> consisting of N-terminal PH domain, a series of four EF hands, a catalytic TIM barrel that the X/Y linker connect its two halves and a C2 domain. | <scene name='70/701452/Fig1/9'>The PLC- β3 has several domains</scene> consisting of N-terminal PH domain, a series of four EF hands, a catalytic TIM barrel that the X/Y linker connect its two halves and a C2 domain. | ||
Revision as of 14:04, 7 July 2015
Unique bidirectional interactions of Phospholipase C beta 3 with G alpha Q
| |||||||||||
References
- ↑ Waldo GL, Ricks TK, Hicks SN, Cheever ML, Kawano T, Tsuboi K, Wang X, Montell C, Kozasa T, Sondek J, Harden TK. Kinetic Scaffolding Mediated by a Phospholipase C-{beta} and Gq Signaling Complex. Science. 2010 Nov 12;330(6006):974-80. Epub 2010 Oct 21. PMID:20966218 doi:10.1126/science.1193438
- ↑ Lyon AM, Tesmer JJ. Structural insights into phospholipase C-beta function. Mol Pharmacol. 2013 Oct;84(4):488-500. doi: 10.1124/mol.113.087403. Epub 2013 Jul, 23. PMID:23880553 doi:http://dx.doi.org/10.1124/mol.113.087403