This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
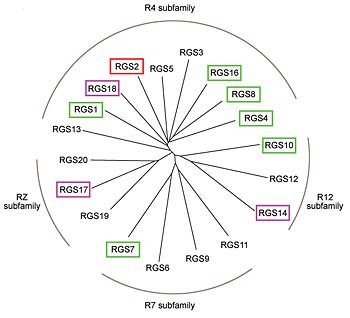
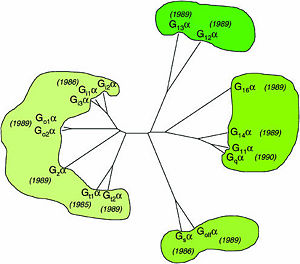
Regulator of G protein signaling
From Proteopedia
(Difference between revisions)
| Line 33: | Line 33: | ||
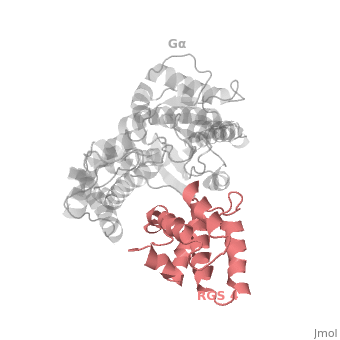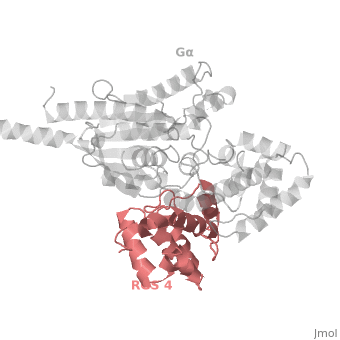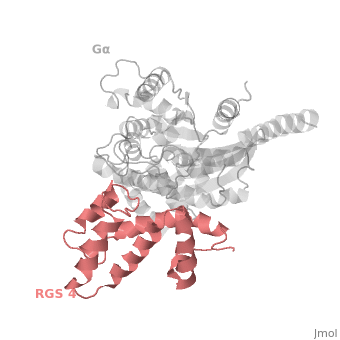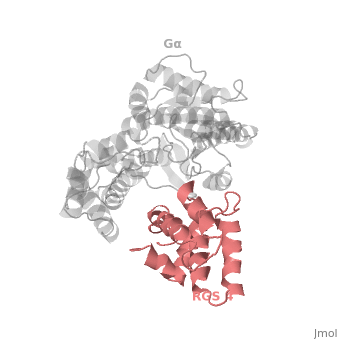
<scene name='70/701447/Rgs4_monomer/5'> The RGS4 domain </scene> corresponds to an array of nine α-helices that fold into two small subdomains, both subdomains are required for it's GAP activity. α1, α2, α3, α4, α5 and α6 helices are colored in blue, aqua, yellow, coral, magenta and dark green respectively while helices α7, α8 and α9 are colored red. | <scene name='70/701447/Rgs4_monomer/5'> The RGS4 domain </scene> corresponds to an array of nine α-helices that fold into two small subdomains, both subdomains are required for it's GAP activity. α1, α2, α3, α4, α5 and α6 helices are colored in blue, aqua, yellow, coral, magenta and dark green respectively while helices α7, α8 and α9 are colored red. | ||
| - | Gα<sub>i1</sub> subunits adopt a conserved fold | + | Gα<sub>i1</sub> subunits adopt a conserved fold of <scene name='70/701447/All-helical-domain/6'>α helical domain</scene> that composed of six α helices shown as blue cartoon, and a conserved GTPase domain shown in gray cartoon. The GTPase domain hydrolyzes GTP and provides most of Gα's binding surfaces for Gβγ, receptors, effectors and RGS proteins. <scene name='70/701447/Gi-rgs4/20'>The GTPase domain</scene> contains three flexible regions designated switch-I presented as blue sticks, switch-II presented as magenta sticks and switch-III presented as green sticks that change conformation in response to GTP binding and hydrolysis, GDP–Mg<sup>+2</sup>, bound in the active site of Gα<sub>i1</sub> is shown as a ball-and-stick model. The three switch regions of Gα<sub>i1</sub>: residues 176–184, 201–215, and 233–241, respectively . <ref>PMID: 9108480</ref> |
== RGS-G proteins interactions == | == RGS-G proteins interactions == | ||
Revision as of 20:31, 25 May 2015
Regulator of G protein signaling (RGS) interactions with G proteins – RGS4-Gαi as a model structure.
| |||||||||||
References
- ↑ Kosloff M, Travis AM, Bosch DE, Siderovski DP, Arshavsky VY. Integrating energy calculations with functional assays to decipher the specificity of G protein-RGS protein interactions. Nat Struct Mol Biol. 2011 Jun 19;18(7):846-53. doi: 10.1038/nsmb.2068. PMID:21685921 doi:http://dx.doi.org/10.1038/nsmb.2068
- ↑ Milligan G, Kostenis E. Heterotrimeric G-proteins: a short history. Br J Pharmacol. 2006 Jan;147 Suppl 1:S46-55. PMID:16402120 doi:http://dx.doi.org/10.1038/sj.bjp.0706405
- ↑ Tesmer JJ, Berman DM, Gilman AG, Sprang SR. Structure of RGS4 bound to AlF4--activated G(i alpha1): stabilization of the transition state for GTP hydrolysis. Cell. 1997 Apr 18;89(2):251-61. PMID:9108480
- ↑ Kosloff M, Travis AM, Bosch DE, Siderovski DP, Arshavsky VY. Integrating energy calculations with functional assays to decipher the specificity of G protein-RGS protein interactions. Nat Struct Mol Biol. 2011 Jun 19;18(7):846-53. doi: 10.1038/nsmb.2068. PMID:21685921 doi:http://dx.doi.org/10.1038/nsmb.2068
Proteopedia Page Contributors and Editors (what is this?)
Ali Asli, Denise Salem, Michal Harel, Joel L. Sussman, Jaime Prilusky