This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Calmodulin
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
==Function== | ==Function== | ||
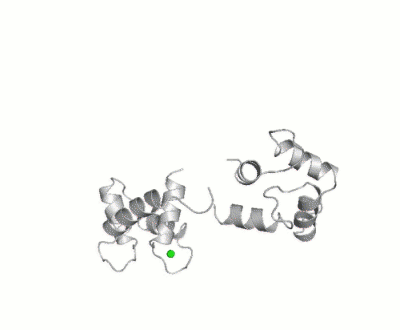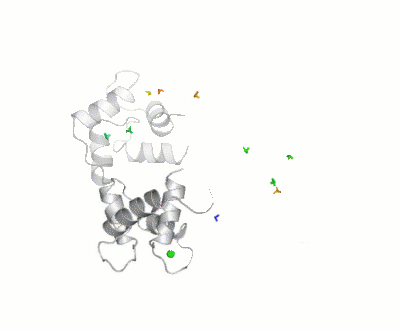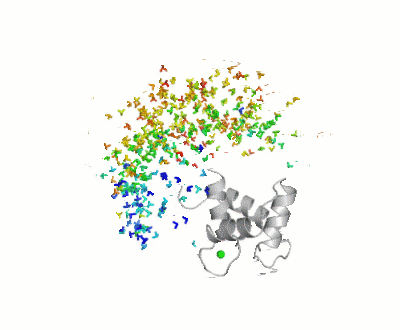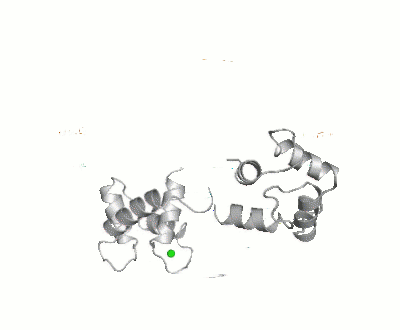
| - | [[Calmodulin]] (CaM) – calcium modulated protein – regulates various protein targets. It is used by various proteins as calcium sensor and signal transducer by binding to their calcium binding domain (CBD). It undergoes conformational change upon binding Ca++ via its 4 [[EF hand]] motives and can undergo post-translational modification. | + | [[Calmodulin]] (CaM) – calcium modulated protein – regulates various protein targets. It is used by various proteins as calcium sensor and signal transducer by binding to their calcium binding domain (CBD). It undergoes conformational change upon binding Ca++ via its 4 [[EF hand]] motives and can undergo post-translational modification. <scene name='39/398280/Cv/4'>Click here to see EF hand</scene> of Human calmodulin (PDB code [[1cll]]). <ref>PMID:1474585</ref> More details on apo-CaM in [[Calcium-free Calmodulin]]. |
{{Clear}} | {{Clear}} | ||
Revision as of 12:20, 18 November 2015
| |||||||||||
3D Structures of Calmodulin
Updated on 18-November-2015
See Also
Bibliography
- ↑ Chattopadhyaya R, Meador WE, Means AR, Quiocho FA. Calmodulin structure refined at 1.7 A resolution. J Mol Biol. 1992 Dec 20;228(4):1177-92. PMID:1474585
- ↑ Bertini I, Giachetti A, Luchinat C, Parigi G, Petoukhov MV, Pierattelli R, Ravera E, Svergun DI. Conformational Space of Flexible Biological Macromolecules from Average Data. J Am Chem Soc. 2010 Sep 7. PMID:20822180 doi:10.1021/ja1063923
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Karsten Theis, Jaime Prilusky, Enrico Ravera, Daniel Moyano-Marino