This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Bacteriorhodopsin
From Proteopedia
(Difference between revisions)
| Line 7: | Line 7: | ||
== Structural highlights == | == Structural highlights == | ||
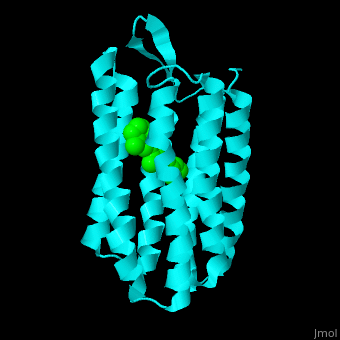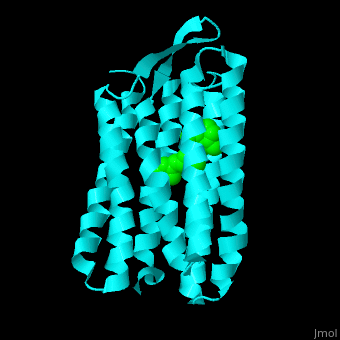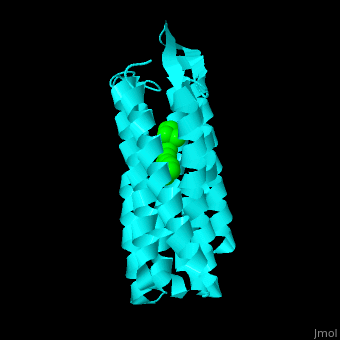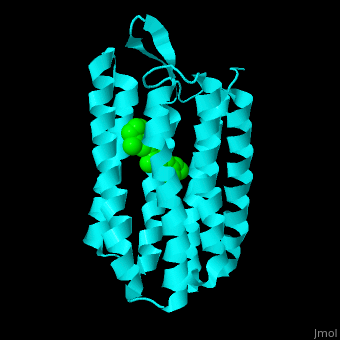
| - | Br is composed of 6 α-helical elements each containing a retinal molecule. The retinal changes its ground state conformation upon binding of a proton, causing the Br to change conformation to the activated state and pump the proton. <scene name='46/463242/Cv/ | + | Br is composed of 6 α-helical elements each containing a retinal molecule. The retinal changes its ground state conformation upon binding of a proton, causing the Br to change conformation to the activated state and pump the proton. <scene name='46/463242/Cv/5'>The retinal interacts with hydrophobic and aromatic residues</scene> <ref>PMID:19603754</ref> ({{Template:ColorKey_Hydrophobic}}, {{Template:ColorKey_Polar}}). |
</StructureSection> | </StructureSection> | ||
== 3D Structures of bacteriorhodopsin == | == 3D Structures of bacteriorhodopsin == | ||
Revision as of 12:04, 30 November 2015
| |||||||||||
3D Structures of bacteriorhodopsin
Updated on 30-November-2015
See Also
References
- ↑ Joh NH, Oberai A, Yang D, Whitelegge JP, Bowie JU. Similar energetic contributions of packing in the core of membrane and water-soluble proteins. J Am Chem Soc. 2009 Aug 12;131(31):10846-7. PMID:19603754 doi:10.1021/ja904711k
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Joel L. Sussman, Wayne Decatur, Jaime Prilusky