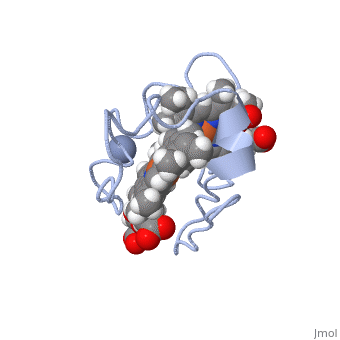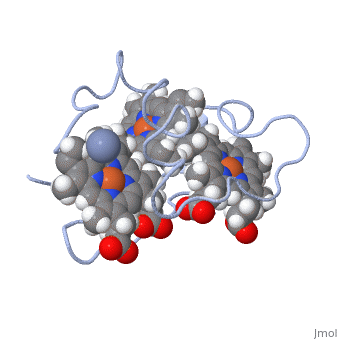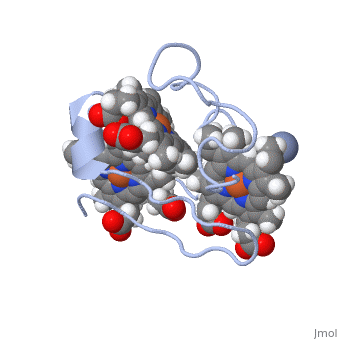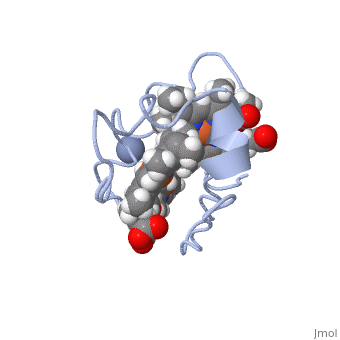This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Cytochrome c 7
From Proteopedia
(Difference between revisions)
| Line 9: | Line 9: | ||
==Structural Components== | ==Structural Components== | ||
| - | Cc7 is a single polypeptide chain 68 residues total containing three heme groups. The polypeptide strand has one alpha helix 4 residues in length and two beta strands 2 residues in length. The heme groups are labelled <scene name='71/716635/Heme_groups/ | + | Cc7 is a single polypeptide chain 68 residues total containing three heme groups. The polypeptide strand has one alpha helix 4 residues in length and two beta strands 2 residues in length. The heme groups are labelled<scene name='71/716635/Heme_groups/2'>as I, III, and IV</scene>. The binding site is located on the distal side of heme group IV where <scene name='71/716635/Active_site_1/1'>lysine residues 41, 42, 46 and 50</scene> are an interactive component of the active site. The net charge of these residues is positive, thus binding to negatively charge compounds or cations<ref name="assfalt" />. |
== Function == | == Function == | ||
Revision as of 18:16, 30 November 2015
General
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Assfalg M, Bertini I, Bruschi M, Michel C, Turano P. The metal reductase activity of some multiheme cytochromes c: NMR structural characterization of the reduction of chromium(VI) to chromium(III) by cytochrome c(7). 2002; 99(15):9750-4 DOI: 10.1073/pnas.152290999
- ↑ Barton L, Fauque G. Biochemistry, Physiology and Biotechnology of Sulfate-Reducing Bacteria. Advances in Applied Microbiology. 2009; 68: 41–98. DOI: 10.1016/s0065-2164(09)01202-7
- ↑ 3.0 3.1 Pfennig N, Biebl H. Desulfuromonas acetoxidans gen. nov. and sp. nov., a new anaerobic, sulfur-reducing, acetate-oxidizing bacterium. 1976; 110(1): 3-12 DOI: 10.1007/BF00416962