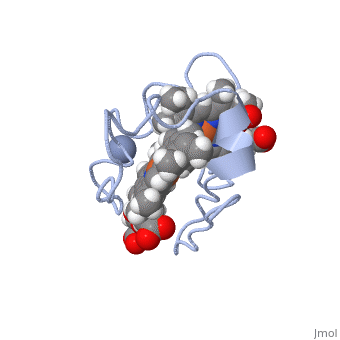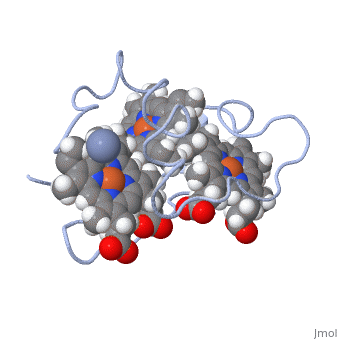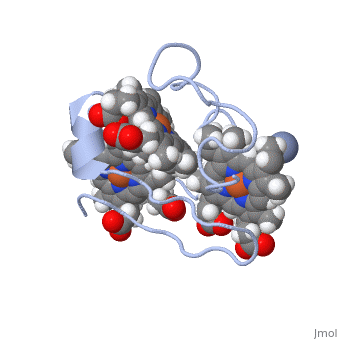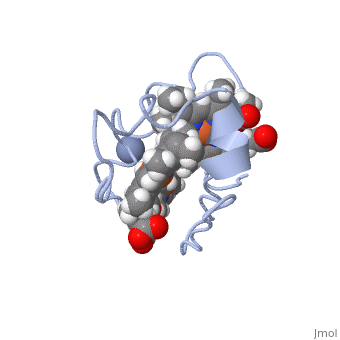This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Cytochrome c 7
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
==General== | ==General== | ||
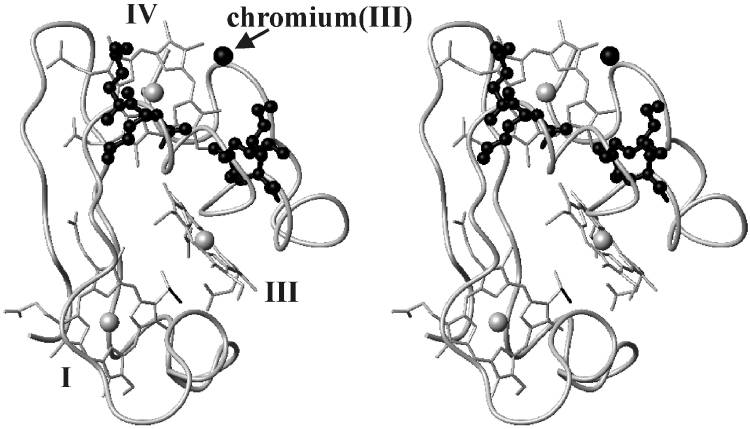
| - | <StructureSection load='1LM2.pdb' size='340' side='right' caption='3D Structure of Cytochrome c | + | <StructureSection load='1LM2.pdb' size='340' side='right' caption='3D Structure of Cytochrome c 7with heme and Cr+3 ion (PDB code [[1lm2]])' scene=''> |
'''Cytocrhome c 7''' (Cc7) is a three heme-containing protein derived from the sulfur-reducing bacterium ''Desulfuromonas acetoxidans''. Cc7 is crucial to the bacteria's anaerobic sulfur respiration as it plays a role in the electron-transfer mechanism, and as such it is located in the mitochondrial inter-membrane space<ref name="assfalt">Assfalg M, Bertini I, Bruschi M, Michel C, Turano P. The metal reductase activity of some multiheme cytochromes c: NMR structural characterization of the reduction of chromium(VI) to chromium(III) by cytochrome c(7). 2002; 99(15):9750-4 '''[http://www.ncbi.nlm.nih.gov/pmc/articles/PMC125002/''' DOI: 10.1073/pnas.152290999''']'''</ref>. Over the course of 3.5 million years, ''Desulfuromonas acetoxidans'' have evolved to use anaerobic sulfur respiration as the main driving force of their survival<ref>Barton L, Fauque G. Biochemistry, Physiology and Biotechnology of Sulfate-Reducing Bacteria. Advances in Applied Microbiology. 2009; 68: 41–98. '''[http://www.sciencedirect.com/science/article/pii/S0065216409012027''' DOI: 10.1016/s0065-2164(09)01202-7''']'''</ref>. | '''Cytocrhome c 7''' (Cc7) is a three heme-containing protein derived from the sulfur-reducing bacterium ''Desulfuromonas acetoxidans''. Cc7 is crucial to the bacteria's anaerobic sulfur respiration as it plays a role in the electron-transfer mechanism, and as such it is located in the mitochondrial inter-membrane space<ref name="assfalt">Assfalg M, Bertini I, Bruschi M, Michel C, Turano P. The metal reductase activity of some multiheme cytochromes c: NMR structural characterization of the reduction of chromium(VI) to chromium(III) by cytochrome c(7). 2002; 99(15):9750-4 '''[http://www.ncbi.nlm.nih.gov/pmc/articles/PMC125002/''' DOI: 10.1073/pnas.152290999''']'''</ref>. Over the course of 3.5 million years, ''Desulfuromonas acetoxidans'' have evolved to use anaerobic sulfur respiration as the main driving force of their survival<ref>Barton L, Fauque G. Biochemistry, Physiology and Biotechnology of Sulfate-Reducing Bacteria. Advances in Applied Microbiology. 2009; 68: 41–98. '''[http://www.sciencedirect.com/science/article/pii/S0065216409012027''' DOI: 10.1016/s0065-2164(09)01202-7''']'''</ref>. | ||
Revision as of 17:42, 5 January 2016
General
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Assfalg M, Bertini I, Bruschi M, Michel C, Turano P. The metal reductase activity of some multiheme cytochromes c: NMR structural characterization of the reduction of chromium(VI) to chromium(III) by cytochrome c(7). 2002; 99(15):9750-4 DOI: 10.1073/pnas.152290999
- ↑ Barton L, Fauque G. Biochemistry, Physiology and Biotechnology of Sulfate-Reducing Bacteria. Advances in Applied Microbiology. 2009; 68: 41–98. DOI: 10.1016/s0065-2164(09)01202-7
- ↑ 3.0 3.1 Pfennig N, Biebl H. Desulfuromonas acetoxidans gen. nov. and sp. nov., a new anaerobic, sulfur-reducing, acetate-oxidizing bacterium. 1976; 110(1): 3-12 DOI: 10.1007/BF00416962