This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Aminomutase
From Proteopedia
(Difference between revisions)
| Line 32: | Line 32: | ||
== Structural highlights == | == Structural highlights == | ||
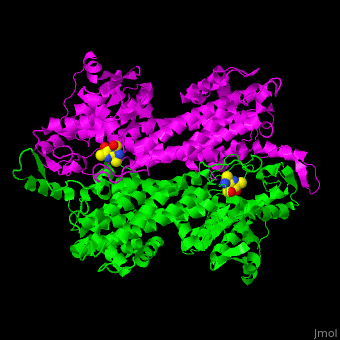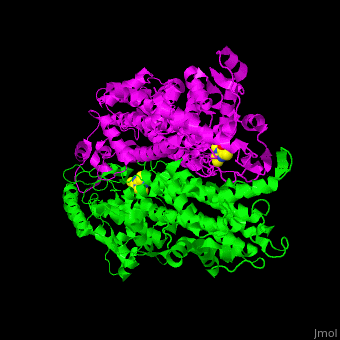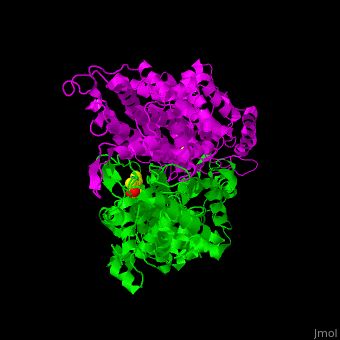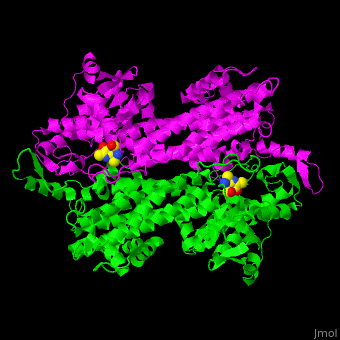
| - | TAM active site residues facilitate the deprotonation/reprotonation reaction. <scene name='71/715468/Cv/2'>The active site</scene> includes residues from both monomers in Tyrosine aminomutase (PDB code [[2ohy]]). | + | TAM active site residues facilitate the deprotonation/reprotonation reaction. <scene name='71/715468/Cv/2'>The active site</scene> includes residues from both monomers in Tyrosine aminomutase (PDB code [[2ohy]]).<ref>PMID:17516659</ref> |
</StructureSection> | </StructureSection> | ||
Revision as of 09:14, 7 December 2015
| |||||||||||
3D structures of aminomutase
Updated on 07-December-2015
References
- ↑ Christianson CV, Montavon TJ, Van Lanen SG, Shen B, Bruner SD. The structure of L-tyrosine 2,3-aminomutase from the C-1027 enediyne antitumor antibiotic biosynthetic pathway. Biochemistry. 2007 Jun 19;46(24):7205-14. Epub 2007 May 22. PMID:17516659 doi:10.1021/bi7003685
- ↑ Christianson CV, Montavon TJ, Van Lanen SG, Shen B, Bruner SD. The structure of L-tyrosine 2,3-aminomutase from the C-1027 enediyne antitumor antibiotic biosynthetic pathway. Biochemistry. 2007 Jun 19;46(24):7205-14. Epub 2007 May 22. PMID:17516659 doi:10.1021/bi7003685