This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
EPSP synthase
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
== Structural insights == | == Structural insights == | ||
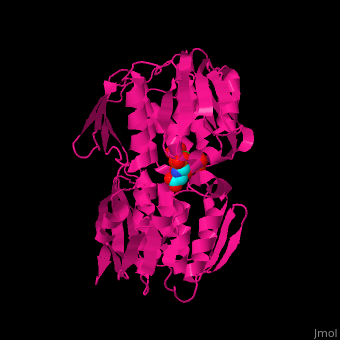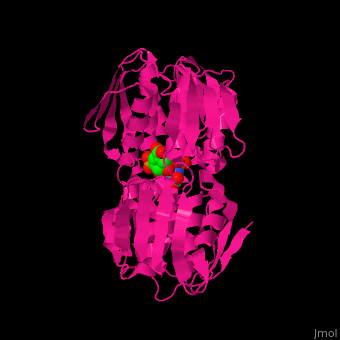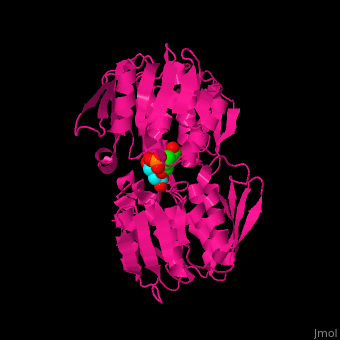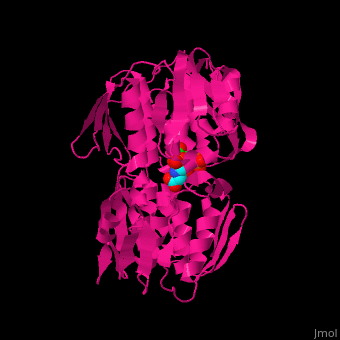
| - | The enzyme has <scene name='57/570585/Two_domains/2'> | + | The enzyme has two domains, with the active site found in the interdomain cleft <scene name='57/570585/Two_domains/2'>(open conformation)</scene>. There is a substantial structural change upon substrate binding, resulting in a <scene name='57/570585/Closed_formation/2'>closed</scene> conformation. <scene name='57/570585/Cv/3'>See animation of this process</scene>. '''Glyphosate''' (also known as '''Roundup''') occupies the <scene name='57/570585/Cv/8'>binding site</scene> of the second substrate, phosphoenol pyruvate <ref>PMID:11171958</ref>. |
</StructureSection> | </StructureSection> | ||
Revision as of 15:54, 20 January 2016
| |||||||||||
3D structures of EPSP synthase
Updated on 20-January-2016
References
- ↑ Priestman MA, Healy ML, Funke T, Becker A, Schonbrunn E. Molecular basis for the glyphosate-insensitivity of the reaction of 5-enolpyruvylshikimate 3-phosphate synthase with shikimate. FEBS Lett. 2005 Oct 24;579(25):5773-80. PMID:16225867 doi:10.1016/j.febslet.2005.09.066
- ↑ Schonbrunn E, Eschenburg S, Shuttleworth WA, Schloss JV, Amrhein N, Evans JN, Kabsch W. Interaction of the herbicide glyphosate with its target enzyme 5-enolpyruvylshikimate 3-phosphate synthase in atomic detail. Proc Natl Acad Sci U S A. 2001 Feb 13;98(4):1376-80. PMID:11171958 doi:http://dx.doi.org/10.1073/pnas.98.4.1376
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Ann Taylor, Joel L. Sussman