This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1137
From Proteopedia
(Difference between revisions)
| Line 9: | Line 9: | ||
=== Molecular effects === | === Molecular effects === | ||
---- | ---- | ||
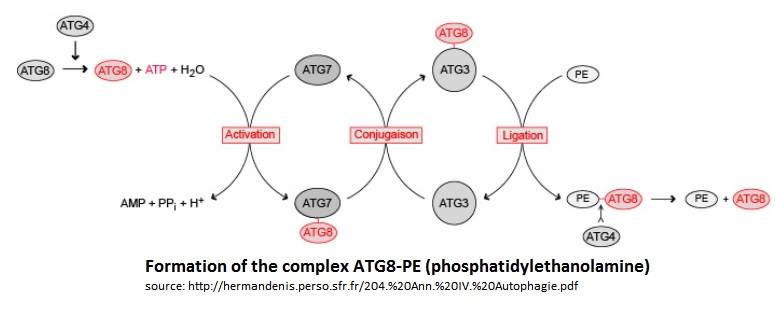
| - | In the Atg8 system, there are several steps until Atg8 is activated. Firstly, the cysteine protease Atg4 cleaves the last residue of the C-terminal domain of Atg8 by consuming 1 molecule of [https://en.wikipedia.org/wiki/Adenosine_triphosphate ATP] and H20. Then, the Atg7 (E1-like enzyme) activates the C-terminal glycine exposed and therefore forms an intermediate Atg8-Atg7 by creating a thioester bond. Atg3 (E2-like enzyme) then replaces Atg7 in the complex to form an Atg8-Atg3 thioester intermediate. Phosphatidylethanolamine (PE) then links to the N-terminal domain of Atg8. This final complex (Atg8-Atg3-PE) serves as an intermediate for membrane tethering and hemifusion of the autophagosome. This complex is primordial for the elongation of the autophagosome. Notice that Atg8-Atg3-PE is the active complex, but Atg8-PE can also be sufficient to do this task (like this is showed on the scheme below). | + | In the Atg8 system, there are several steps until Atg8 is activated. Firstly, the cysteine protease Atg4 cleaves the last residue of the C-terminal domain of Atg8 by consuming 1 molecule of [https://en.wikipedia.org/wiki/Adenosine_triphosphate ATP] and H20. Then, the Atg7 (E1-like enzyme) activates the C-terminal glycine exposed and therefore forms an intermediate Atg8-Atg7 by creating a thioester bond. Atg3 (E2-like enzyme) then replaces Atg7 in the complex to form an Atg8-Atg3 thioester intermediate.[https://fr.wikipedia.org/wiki/Phosphatidyl%C3%A9thanolamine Phosphatidylethanolamine] (PE) then links to the N-terminal domain of Atg8. This final complex (Atg8-Atg3-PE) serves as an intermediate for membrane tethering and hemifusion of the autophagosome. This complex is primordial for the elongation of the autophagosome. Notice that Atg8-Atg3-PE is the active complex, but Atg8-PE can also be sufficient to do this task (like this is showed on the scheme below). |
[[Image:atg8f.jpg]] | [[Image:atg8f.jpg]] | ||
Revision as of 19:56, 29 January 2016
| This Sandbox is Reserved from 15/12/2015, through 15/06/2016 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1120 through Sandbox Reserved 1159. |
To get started:
More help: Help:Editing |
Plasmodium falciparum Atg8 in complex with Plasmodium falciparum Atg3 peptide
| |||||||||||
References
http://hermandenis.perso.sfr.fr/204.%20Ann.%20IV.%20Autophagie.pdf