This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox HEC
From Proteopedia
(Difference between revisions)
| Line 12: | Line 12: | ||
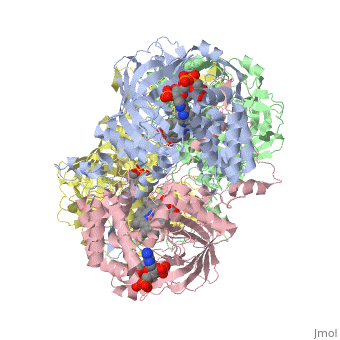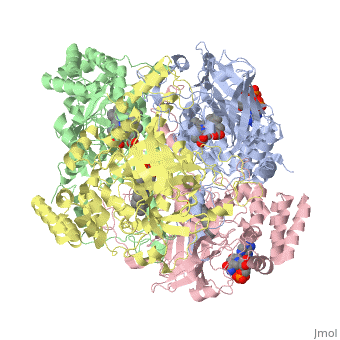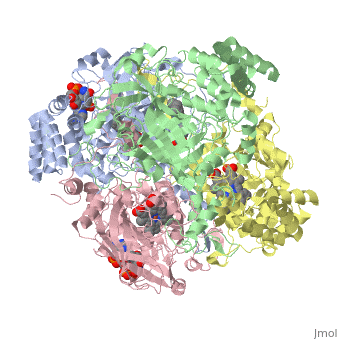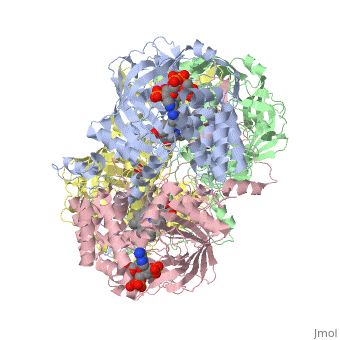
Human erythrocyte catalase is a negatively charged heme-containing monofunctional tetrameric enzyme prevalent among aerobic organisms <ref name= Kodydková >PMID:25152049</ref><ref name=Alfonso-Prietro>PMID:22516655</ref><ref name=Dash>PMID:22521743</ref><ref name=Diaz>PMID:22209752 </ref><ref name=Nishikawa>PMID:19385054 </ref>([[Kodydková, Vávrová, Kocík, & Zák, A., 2014; Alfonso-Prietro, Vidossich, & Rovira, 2012; Dash & Phillips, 2012; Diaz, Loewen, Fita, & Carpena, 2012; Nishikawa, Hashida, & Takakura, 2009)]]. Stable forms of hydrogen peroxide are beneficial in biological reactions including hypoxia signal transduction, cell proliferation and differentiation regulation, and immune response mediation; however, it is toxic at high levels as free hydroxyl ions cannot be catalyzed by the body <ref name= Lennicke >PMID:26369938</ref><ref name= "halliwell">DOI: 10.1016/S0014-5739(00)02197</ref> ([[Lennicke et al., 2015]]; Halliwell, Clement, & Long, 2000). Within this catalytic group, hydrogen peroxide acts to both oxidize and reduce the reaction. Catalase ultimately functions to break down hydrogen peroxide (Dash & Phillips, 2012). This is accomplished in a two-step mechanism where the heme is first oxidized by a molecule of hydrogen peroxide to produce Compound I, a high energy oxyferryl cation radical intermediate, as well as a water molecule. Compound I is then immediately reduced by a second hydrogen peroxide molecule to produce a second molecule of water (Alfonso-Prietro, Vidossich, & Rovira, 2012; Diaz, Loewen, Fita, & Carpena, 2012). The overall reaction results in two single-electron removal transfers from the iron atom of the heme group and the porphyrin from the oxoferryl radical, and a proton transfer from histidine. The mechanism is enthalpically driven by the distal histidine proton transfer as it is more exothermic than the electron transfers (Alfonso-Prietro, Vidossich, & Rovira, 2012; Diaz, Loewen, Fita, & Carpena, 2012). | Human erythrocyte catalase is a negatively charged heme-containing monofunctional tetrameric enzyme prevalent among aerobic organisms <ref name= Kodydková >PMID:25152049</ref><ref name=Alfonso-Prietro>PMID:22516655</ref><ref name=Dash>PMID:22521743</ref><ref name=Diaz>PMID:22209752 </ref><ref name=Nishikawa>PMID:19385054 </ref>([[Kodydková, Vávrová, Kocík, & Zák, A., 2014; Alfonso-Prietro, Vidossich, & Rovira, 2012; Dash & Phillips, 2012; Diaz, Loewen, Fita, & Carpena, 2012; Nishikawa, Hashida, & Takakura, 2009)]]. Stable forms of hydrogen peroxide are beneficial in biological reactions including hypoxia signal transduction, cell proliferation and differentiation regulation, and immune response mediation; however, it is toxic at high levels as free hydroxyl ions cannot be catalyzed by the body <ref name= Lennicke >PMID:26369938</ref><ref name= "halliwell">DOI: 10.1016/S0014-5739(00)02197</ref> ([[Lennicke et al., 2015]]; Halliwell, Clement, & Long, 2000). Within this catalytic group, hydrogen peroxide acts to both oxidize and reduce the reaction. Catalase ultimately functions to break down hydrogen peroxide (Dash & Phillips, 2012). This is accomplished in a two-step mechanism where the heme is first oxidized by a molecule of hydrogen peroxide to produce Compound I, a high energy oxyferryl cation radical intermediate, as well as a water molecule. Compound I is then immediately reduced by a second hydrogen peroxide molecule to produce a second molecule of water (Alfonso-Prietro, Vidossich, & Rovira, 2012; Diaz, Loewen, Fita, & Carpena, 2012). The overall reaction results in two single-electron removal transfers from the iron atom of the heme group and the porphyrin from the oxoferryl radical, and a proton transfer from histidine. The mechanism is enthalpically driven by the distal histidine proton transfer as it is more exothermic than the electron transfers (Alfonso-Prietro, Vidossich, & Rovira, 2012; Diaz, Loewen, Fita, & Carpena, 2012). | ||
| - | The catalase fold, a stereoscopic alignment of the clade 3 subunits, contains an eight-sheeted anti-parallel beta-barrel domain linked to a six alpha-helical domain via a lengthy protein sequence. Residues within β1-β4 contribute to the heme variant, while monomers within β5-β8 establish the NADPH binding site (Diaz, Loewen, Fita, & Carpena, 2012). The positioning of the heme is determined by the proximal aromatic pyrrole compounds <scene name='3cs9/Overall_structure/1'>TextToBeDisplayed</scene>; in human erythrocyte catalase, catalytic His75 is positioned above pyrrole ring III, further producing a His-III orientation and heme-b variant. The NADPH binding site is located at the β,α-domain junction (Alfonso-Prietro, Vidossich, & Rovira, 2012; Diaz, Loewen, Fita, & Carpena, 2012). When the NADPH molecule is bound, a right-handed clockwise helical formation is produced. In human erythrocyte catalase, only two of the four subunits allow for NADPH binding | + | The catalase fold, a stereoscopic alignment of the clade 3 subunits, contains an eight-sheeted anti-parallel beta-barrel domain linked to a six alpha-helical domain via a lengthy protein sequence. Residues within β1-β4 contribute to the heme variant, while monomers within β5-β8 establish the NADPH binding site (Diaz, Loewen, Fita, & Carpena, 2012). The positioning of the heme is determined by the proximal aromatic pyrrole compounds <scene name='3cs9/Overall_structure/1'>TextToBeDisplayed</scene>; in human erythrocyte catalase, catalytic His75 is positioned above pyrrole ring III, further producing a His-III orientation and heme-b variant. The NADPH binding site is located at the β,α-domain junction (Alfonso-Prietro, Vidossich, & Rovira, 2012; Diaz, Loewen, Fita, & Carpena, 2012). When the NADPH molecule is bound, a right-handed clockwise helical formation is produced. In human erythrocyte catalase, only two of the four subunits allow for NADPH binding (Kodydková, Vávrová, Kocík, & Zák, A., 2014; Diaz, Loewen, Fita, & Carpena, 2012). The active site contains a negatively charged tyrosine and a positively charged histidine situated, respectively, proximal and distal to the heme group. The histidine is responsible for the formation of Compound I during the first step of the catalase mechanism (Alfonso-Prietro, Vidossich, & Rovira, 2012). |
The deeply buried heme group is connected to the protein surface by a primary channel which provides a transport pathway for the hydrogen peroxide substrate (Diaz, Loewen, Fita, & Carpena, 2012). The transportation of hydrogen peroxide through the main channel is regulated by electrical dipole interactions between the hydrogen peroxide and the hydrophobic portion of the channel containing negatively charged aspartate and positively charged iron from the heme (Lennicke et al., 2015; Diaz, Loewen, Fita, & Carpena, 2012; Halliwell, Clement, & Long, 2000). Additionally, less significant lateral channels allow products to leave the heme pocket (Diaz, Loewen, Fita, & Carpena, 2012). Human erythrocyte catalase is not evenly distributed throughout the body due to restricted endothelium passageways; this allows for a controlled and localized dissemination of the protein (Nishikawa, Hashida, & Takakura, 2009). | The deeply buried heme group is connected to the protein surface by a primary channel which provides a transport pathway for the hydrogen peroxide substrate (Diaz, Loewen, Fita, & Carpena, 2012). The transportation of hydrogen peroxide through the main channel is regulated by electrical dipole interactions between the hydrogen peroxide and the hydrophobic portion of the channel containing negatively charged aspartate and positively charged iron from the heme (Lennicke et al., 2015; Diaz, Loewen, Fita, & Carpena, 2012; Halliwell, Clement, & Long, 2000). Additionally, less significant lateral channels allow products to leave the heme pocket (Diaz, Loewen, Fita, & Carpena, 2012). Human erythrocyte catalase is not evenly distributed throughout the body due to restricted endothelium passageways; this allows for a controlled and localized dissemination of the protein (Nishikawa, Hashida, & Takakura, 2009). | ||
| Line 18: | Line 18: | ||
== Disease and Disorders == | == Disease and Disorders == | ||
| - | There are 12 known mutations in the human erythrocyte catalase gene that have been found to cause acatalasemia (5). Acatalasemia is an autosomal recessive condition in which human erythrocyte catalase levels are very low. Most people are asymptomatic and are diagnosed because a family member is affected. However, although they are asymptomatic, they have an increased risk of chronic diseases. | + | There are 12 known mutations in the human erythrocyte catalase gene that have been found to cause acatalasemia <ref name="László Góth">PMID:22365890</ref> (5). Acatalasemia is an autosomal recessive condition in which human erythrocyte catalase levels are very low. Most people are asymptomatic and are diagnosed because a family member is affected. However, although they are asymptomatic, they have an increased risk of chronic diseases. |
| - | Acatalasemia can be correlated with ulcers and gangrene. When this occurs, the condition is known as Takahara disease. Ulcers and gangrene can result from high levels of hydrogen peroxide that is normally produced from bacteria. Mutations in the human erythrocyte catalase gene tend to reduce the activity of human erythrocyte catalase (1) to less than 10% of its normal activity thus reducing the enzymes ability to degrade hydrogen peroxide and causing a build-up of hydrogen peroxide. This build-up in turn causes ulcers and gangrene. | + | Acatalasemia can be correlated with ulcers and gangrene. When this occurs, the condition is known as Takahara disease. Ulcers and gangrene can result from high levels of hydrogen peroxide that is normally produced from bacteria. Mutations in the human erythrocyte catalase gene tend to reduce the activity of human erythrocyte catalase <ref name="Góth">PMID:24025477</ref> (1) to less than 10% of its normal activity thus reducing the enzymes ability to degrade hydrogen peroxide and causing a build-up of hydrogen peroxide. This build-up in turn causes ulcers and gangrene. |
| - | A similar condition to acatalasemia is hypocatalasemia, in which each cell of the human erythrocyte catalase only has one gene with a mutation, instead of both genes with a mutation. This single mutation cuts the activity of human erythrocyte catalase by about half. Similar to acatalasemia, this condition normally doesn’t cause health issues (2). | + | A similar condition to acatalasemia is hypocatalasemia, in which each cell of the human erythrocyte catalase only has one gene with a mutation, instead of both genes with a mutation. This single mutation cuts the activity of human erythrocyte catalase by about half. Similar to acatalasemia, this condition normally doesn’t cause health issues <ref name="Góth,Eaton">PMID:11117918</ref>(2). |
Acatalasemia is also associated with type 2 diabetes mellitus, the most common form of diabetes. The build-up of hydrogen peroxide from the decrease in human erythrocyte catalase can damage beta cells in the pancreas. The pancreas releases insulin, which helps your body regulate your blood sugar level. However, the damaged beta cells cannot utilize the insulin as well as normal beta cells, which leads to type 2 diabetes mellitus. These defective beta cells are thought to be why people with acatalasemia have an increased risk for type 2 diabetes mellitus. A larger percentage of people with diabetes have acatalasemia than those with diabetes without acatalasemia. Those with acatalasemia also tend to develop diabetes at an earlier age (1). | Acatalasemia is also associated with type 2 diabetes mellitus, the most common form of diabetes. The build-up of hydrogen peroxide from the decrease in human erythrocyte catalase can damage beta cells in the pancreas. The pancreas releases insulin, which helps your body regulate your blood sugar level. However, the damaged beta cells cannot utilize the insulin as well as normal beta cells, which leads to type 2 diabetes mellitus. These defective beta cells are thought to be why people with acatalasemia have an increased risk for type 2 diabetes mellitus. A larger percentage of people with diabetes have acatalasemia than those with diabetes without acatalasemia. Those with acatalasemia also tend to develop diabetes at an earlier age (1). | ||
Revision as of 23:10, 7 April 2016
1dgb
| |||||||||||