This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Nos1
From Proteopedia
| Line 22: | Line 22: | ||
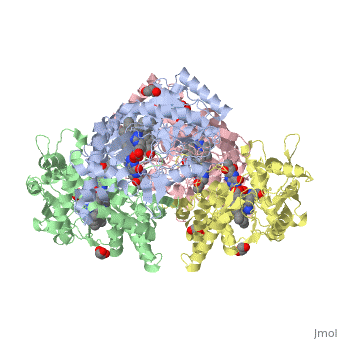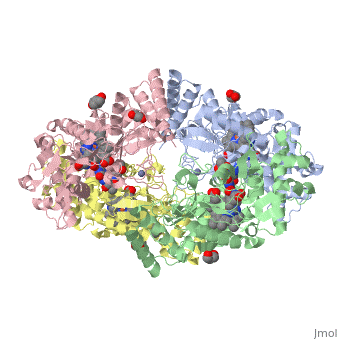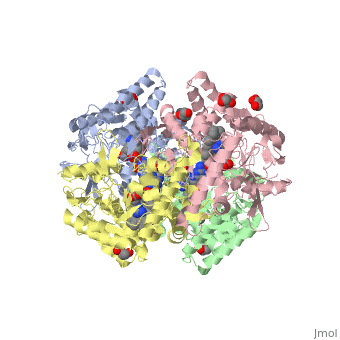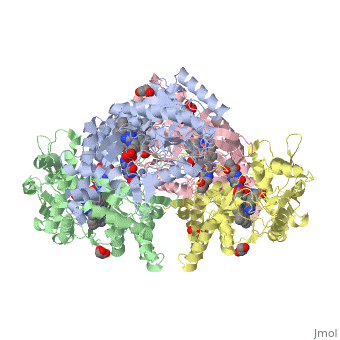
The Nitric oxide synthase gene exists as a homodimer. This homodimer consists of two regions. One region is an N-terminal oxygenase domain and the other is a C-terminal reductase. The N-terminal oxygenase domain is an extended beta sheet cage with binding sites for heme. The N-terminal catalytic domain contains a <scene name='72/728110/Heme_active_site/1'>heme active site</scene> , a nearby cofactor site for tetrahydrobiopterin ( <scene name='72/728110/H4b/1'>H4b</scene> ) and a C-terminal reductase domain that consists of FMN, FAD, and NADPH binding sites. The structure of the human nNOS heme domain contains cysteine and tryptophan. The NADPH provides electrons for catalysis. These electrons are first passed to FAD and FMN and then the heme. This electron flow is monitored by the binding of CaM and Calcium2+ in the linker region between the two heterodimer domains. | The Nitric oxide synthase gene exists as a homodimer. This homodimer consists of two regions. One region is an N-terminal oxygenase domain and the other is a C-terminal reductase. The N-terminal oxygenase domain is an extended beta sheet cage with binding sites for heme. The N-terminal catalytic domain contains a <scene name='72/728110/Heme_active_site/1'>heme active site</scene> , a nearby cofactor site for tetrahydrobiopterin ( <scene name='72/728110/H4b/1'>H4b</scene> ) and a C-terminal reductase domain that consists of FMN, FAD, and NADPH binding sites. The structure of the human nNOS heme domain contains cysteine and tryptophan. The NADPH provides electrons for catalysis. These electrons are first passed to FAD and FMN and then the heme. This electron flow is monitored by the binding of CaM and Calcium2+ in the linker region between the two heterodimer domains. | ||
The amino acid composition of asparagine and methionine in rat neuronal NOS are conserved in human nNOS. This amino acid composition influences the binding of inhibitors and makes nNos inhibitors highly selective. The structure of the human nNOS heme domain contains cysteine and tryptophan. The cysteine subunits of one region pack closely against the backbone of histidine in subunits of another region. Crystal packing interactions most likely contribute to the fully ordered N-termini in the NOS structure | The amino acid composition of asparagine and methionine in rat neuronal NOS are conserved in human nNOS. This amino acid composition influences the binding of inhibitors and makes nNos inhibitors highly selective. The structure of the human nNOS heme domain contains cysteine and tryptophan. The cysteine subunits of one region pack closely against the backbone of histidine in subunits of another region. Crystal packing interactions most likely contribute to the fully ordered N-termini in the NOS structure | ||
| - | There is a flexible surface loop downstream from the Zn2+ binding site. This loop in human nNOS lacks any secondary structure and appears as a random coil. This reflects the flexible nature of nNOS because it can easily adapt to local environment which involves crystal packing. This loop is found near the CaM binding site and can therefore most likely interact with it and affect electron flow. | + | There is a flexible surface loop downstream from the <scene name='72/728110/Zinc/1'>Zn2+</scene> binding site. This loop in human nNOS lacks any secondary structure and appears as a random coil. This reflects the flexible nature of nNOS because it can easily adapt to local environment which involves crystal packing. This loop is found near the CaM binding site and can therefore most likely interact with it and affect electron flow. |
Revision as of 23:59, 27 April 2016
| |||||||||||
References
Shinkai, T., Ohmori, O., Hori, H., and Nakamura, J. (2002) Allelic association of the neuronal nitric oxide synthase (NOS1) gene with schizophrenia. Molecular Psychiatry. 7, 560-563. doi:10.1038/sj.mp.4001041 Galimberti, D., Scarpini, E., Venturelli, E., Strobel, A., Herterich, S., Fenogolio, C., Guidi, I., Scalabrini, D., Cortini, F., Bresolin, N., Lesch, K., and Reif, A. (2008) Association of a NOS1 promoter repeat with Alzheimer’s disease. Neurobiology of Aging. 29, 1359-1365. doi:10.1016/j.neurobiolaging.2007.03.003 Rife, T., Rasoul, B., Pullen, N., Mitchell, D., Grathwol, K., and Kurth, J. (2009) The effect of a promoter polymorphism on transcription of nitric oxide synthase 1 and its relevance to Parkinson’s disease. Journal of Neuroscience Research. 87, 2319-2325. doi:10.1005/jnr.22045
- ↑ UniProt Consortium 2009, ‘UniProtKB - P29475 (NOS1_HUMAN),’ UniProtKB Protein Knowledgebase
- ↑ Ward ME, Toporsian M, Scott JA, et al. Hypoxia induces a functionally significant and translationally efficient neuronal NO synthase mRNA variant. Journal of Clinical Investigation. 2005;115(11):3128-3139. doi:10.1172/JCI20806.
- ↑ Juliane Kopf, Martin Schecklmann, Tim Hahn, Thomas Dresler, Alica C. Dieler, Martin J. Herrmann, Andreas J. Fallgatter, Andreas Reif. (2011) NOS1 ex1f-VNTR polymorphism influences prefrontal brain oxygenation during a working memory task, NeuroImage, Volume 57, (Issue 4),1617-1623, Ihttp://dx.doi.org/10.1016/
- ↑ Freudenberg, F., Alttoa, A. & Reif, A. (2015) Neuronal nitric oxide synthase (NOS1) and its adaptor (NOS1AP) act as a genetic risk factors for psychiatric. Genes Brain Behav 14, 47–64.
- ↑ Zhang, Y. H., Jin, C. Z., Jang, J. H., & Wang, Y. (2014). Molecular mechanisms of neuronal nitric oxide synthase in cardiac function and pathophysiology. The Journal of Physiology, 592(Pt 15), 3189–3200. http://doi.org/10.1113/jphysiol.2013.270306]
- ↑ Zhang, Y. H., Jin, C. Z., Jang, J. H., & Wang, Y. (2014). Molecular mechanisms of neuronal nitric oxide synthase in cardiac function and pathophysiology. The Journal of Physiology, 592(Pt 15), 3189–3200. http://doi.org/10.1113/jphysiol.2013.270306]
- ↑ Zhang, Y. H., Jin, C. Z., Jang, J. H., & Wang, Y. (2014). Molecular mechanisms of neuronal nitric oxide synthase in cardiac function and pathophysiology. The Journal of Physiology, 592(Pt 15), 3189–3200. http://doi.org/10.1113/jphysiol.2013.270306]
Li H, Jamal J, Plaza C, et al. Structures of human constitutive nitric oxide synthases. Acta Crystallographica Section D: Biological Crystallography. 2014;70(Pt 10):2667-2674.