This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:R. Jeremy Johnson/GPR40
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
== Background == | == Background == | ||
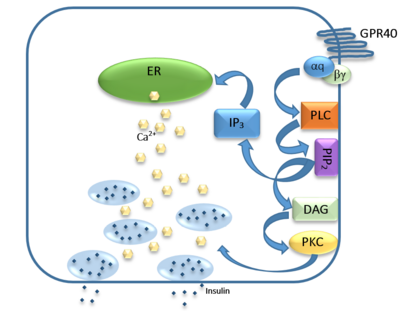
| - | Human G-protein coupled receptor 40 (hGPR40), also known as free fatty acid 1 receptor (FFAR1), is a seven helical transmembrane domain receptor for long-chain free [https://en.wikipedia.org/wiki/Fatty_acid fatty acids] that induces insulin secretion.<ref name="Srivastava">PMID:25043059</ref> Some known fatty acid substrates of hGPR40 include [http://www.news-medical.net/health/What-is-Linoleic-Acid.aspx linoleic acid], [http://www.livestrong.com/article/438717-what-is-oleic-acid/ oleic acid], [http://www.hmdb.ca/metabolites/hmdb02925 eicosatrienoic acid], and [https://en.wikipedia.org/wiki/Palmitoleic_acid palmitoleic acid]<ref name="Morgan">PMID:19660440</ref>. hGPR40 is highly expressed in human pancreatic [https://en.wikipedia.org/wiki/Beta_cell β cells], brain, and endocrine cells of the [https://en.wikipedia.org/wiki/Gastrointestinal_tract gastrointestinal tract] <ref name=”REN”>PMID:26974599</ref>. hGPR40 is of particular interest because the triggering of insulin secrection is [https://en.wikipedia.org/wiki/Glucose glucose] dependent.This glucose-dependence for hGPR40 signaling makes it a target for the treatment of [https://en.wikipedia.org/wiki/Diabetes_mellitus_type_2 type-2 diabetes] as agonists could increase glycemic control and lower the risk of hypoglycemia<ref name=" | + | Human G-protein coupled receptor 40 (hGPR40), also known as free fatty acid 1 receptor (FFAR1), is a seven helical transmembrane domain receptor for long-chain free [https://en.wikipedia.org/wiki/Fatty_acid fatty acids] that induces insulin secretion.<ref name="Srivastava">PMID:25043059</ref> Some known fatty acid substrates of hGPR40 include [http://www.news-medical.net/health/What-is-Linoleic-Acid.aspx linoleic acid], [http://www.livestrong.com/article/438717-what-is-oleic-acid/ oleic acid], [http://www.hmdb.ca/metabolites/hmdb02925 eicosatrienoic acid], and [https://en.wikipedia.org/wiki/Palmitoleic_acid palmitoleic acid]<ref name="Morgan">PMID:19660440</ref>. hGPR40 is highly expressed in human pancreatic [https://en.wikipedia.org/wiki/Beta_cell β cells], brain, and endocrine cells of the [https://en.wikipedia.org/wiki/Gastrointestinal_tract gastrointestinal tract] <ref name=”REN”>PMID:26974599</ref>. hGPR40 is of particular interest because the triggering of insulin secrection is [https://en.wikipedia.org/wiki/Glucose glucose] dependent.This glucose-dependence for hGPR40 signaling makes it a target for the treatment of [https://en.wikipedia.org/wiki/Diabetes_mellitus_type_2 type-2 diabetes] as agonists could increase glycemic control and lower the risk of hypoglycemia.<ref name="Srivastava"/> GPR40 is a member of a group of homologous [[GPCRs]] all located on chromosome 19q13.1 including GPCR41, 42, and 43.<ref name="Burant">PMID:23882043</ref> Evidence exists that shows GPCR43 is involved in adipogenesis. |
== Function == | == Function == | ||
| Line 13: | Line 13: | ||
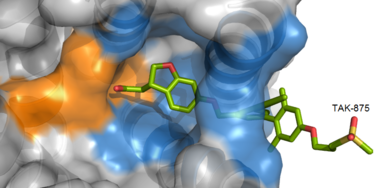
Like most G-protein coupled receptors, hGPR40 contains <scene name='72/721541/Top_view_transmembrane_helices/2'>seven transmembrane helices</scene> (<scene name='72/721541/Top_view_transmembrane_helices/1'>top view of TM helices</scene>). To obtain a [https://en.wikipedia.org/wiki/Protein_crystallization crystallized structure] of the protein, four <scene name='72/721541/Stabilizing_mutations/4'>stabilizing mutations</scene> (<scene name='72/721541/L42a/3'>L42A</scene>, <scene name='72/721541/F88a/4'>F88A</scene>, <scene name='72/721541/G103a/3'>G103A</scene>, <scene name='72/721541/Y202f/3'>Y202F</scene>) were made to increase expression levels and thermal stability of the protein. These mutations did not significantly impact the enzyme's binding affinity with a known agonist, TAK-875.<ref name="Srivastava"/> A <scene name='72/721541/Lysozyme_crimson/2'>T4 Lysozyme</scene> (shown in <FONT COLOR="#DC143C">'''crimson'''</FONT>) was also added to intracellular loop 3 to aid in the formation of crystals. T4 Lysozyme had little effect on TAK-875 binding.<ref name="Srivastava"/> For clarity, lysozyme is removed in all further renderings of hGPR40. hGPR40 also contains an extracellular loop that is conserved among most G-protein coupled receptors (ECL2). This loop has two subsections and is involved in the permeability of the binding site. | Like most G-protein coupled receptors, hGPR40 contains <scene name='72/721541/Top_view_transmembrane_helices/2'>seven transmembrane helices</scene> (<scene name='72/721541/Top_view_transmembrane_helices/1'>top view of TM helices</scene>). To obtain a [https://en.wikipedia.org/wiki/Protein_crystallization crystallized structure] of the protein, four <scene name='72/721541/Stabilizing_mutations/4'>stabilizing mutations</scene> (<scene name='72/721541/L42a/3'>L42A</scene>, <scene name='72/721541/F88a/4'>F88A</scene>, <scene name='72/721541/G103a/3'>G103A</scene>, <scene name='72/721541/Y202f/3'>Y202F</scene>) were made to increase expression levels and thermal stability of the protein. These mutations did not significantly impact the enzyme's binding affinity with a known agonist, TAK-875.<ref name="Srivastava"/> A <scene name='72/721541/Lysozyme_crimson/2'>T4 Lysozyme</scene> (shown in <FONT COLOR="#DC143C">'''crimson'''</FONT>) was also added to intracellular loop 3 to aid in the formation of crystals. T4 Lysozyme had little effect on TAK-875 binding.<ref name="Srivastava"/> For clarity, lysozyme is removed in all further renderings of hGPR40. hGPR40 also contains an extracellular loop that is conserved among most G-protein coupled receptors (ECL2). This loop has two subsections and is involved in the permeability of the binding site. | ||
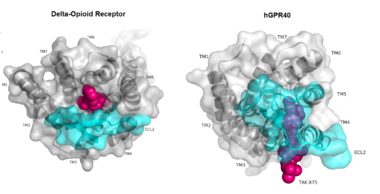
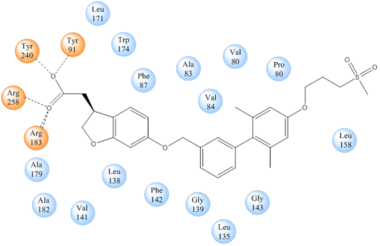
| - | While there is relatively low sequence identity between hGPR40 and peptide-binding and [https://en.wikipedia.org/wiki/Opioid_receptor opioid GPCRs], they do share structural similarities such as a conserved <scene name='72/727085/Hairpin_loop/4'>hairpin loop</scene> motif on <scene name='72/727085/Ecl2/4'>extracellular loop 2 </scene>(ECL2)<ref name=" | + | While there is relatively low sequence identity between hGPR40 and peptide-binding and [https://en.wikipedia.org/wiki/Opioid_receptor opioid GPCRs], they do share structural similarities such as a conserved <scene name='72/727085/Hairpin_loop/4'>hairpin loop</scene> motif on <scene name='72/727085/Ecl2/4'>extracellular loop 2 </scene>(ECL2).<ref name="Srivastava"/> In addition, there is a conserved <scene name='72/727085/Disulfide/3'>disulphide bond</scene> that is formed between transmembrane helix 3 (Cys 79) and the C-terminus of ECL2 (Cys170).<ref name="Srivastava"/> Compared to peptide-binding and opioid GPCRs which have distinctive [https://en.wikipedia.org/wiki/Beta_sheet β-sheets] spanning from transmembrane helix 4 to 5, hGPR40 possesses a shorter B-sheet-like region which has [http://proteopedia.org/wiki/index.php/Image:Beta-like_factors_of_hGPR40_ECL2.png low B-factors].<ref name="Srivastava"/> This reflects the low mobility of the region that limits the overall flexibility of the adjacent portion of ECL2 between Leu171 and Asp175.<ref name="Srivastava"/> A unique feature of hGPR40 is the presence of an additional 13 residues (Pro147 to Gly159) on ECL2 which is absent on all the other peptide/opioid receptors.<ref name="Srivastava"/> These extra residues form a separate <scene name='72/727085/Auxiliary_loop/3'>auxiliary loop</scene> between the B-sheet-like region and transmembrane 4. Together, the auxiliary loop and ECL2 of hGPR40 function as a <scene name='72/727085/Ecl2_cap/3'>roof </scene> over the canonical binding site covering it from the central extracellular region.<ref name="Srivastava"/> |
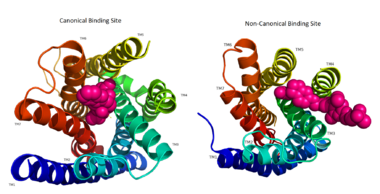
| - | The canonical binding pocket for many other GPCRs is solvent exposed and centrally located between the transmembrane helices allowing ligands to directly bind from the extracellular space<ref name=" | + | The canonical binding pocket for many other GPCRs is solvent exposed and centrally located between the transmembrane helices allowing ligands to directly bind from the extracellular space.<ref name="Srivastava"/> However, because the ECL2 acts as a roof to this canonical binding site, it inhibits ligands from entering directly from the extracellular region. Instead, the highly lipophilic nature of hGPRC40’s ligands allow it to enter a <scene name='72/727085/Hgpr40_entry/2'>noncanonical binding pocket </scene> between TM3 and TM4 by moving through the lipid bilayer.<ref name="Srivastava"/> |
[[Image:Binding site comparison.png|380 px|thumb|center|'''Figure 2:''' Comparison of the canonical binding site represented in pink of most opioid/peptide binding GPCRs (left) compared to the noncanonical binding site of ligands with hGPR40 (right). ]] | [[Image:Binding site comparison.png|380 px|thumb|center|'''Figure 2:''' Comparison of the canonical binding site represented in pink of most opioid/peptide binding GPCRs (left) compared to the noncanonical binding site of ligands with hGPR40 (right). ]] | ||
Revision as of 19:55, 9 June 2016
Human GPR40 (hGPR40), also known as Free Fatty Acid Receptor 1 (FFAR1)
| |||||||||||