This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
TNF receptor-associated factor
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
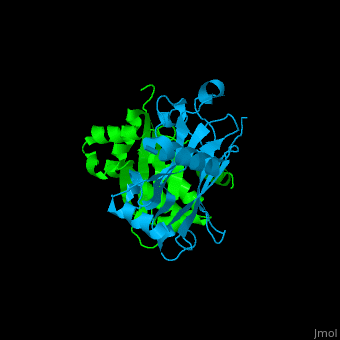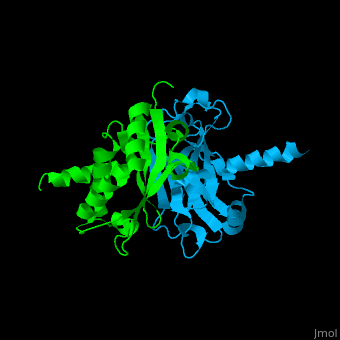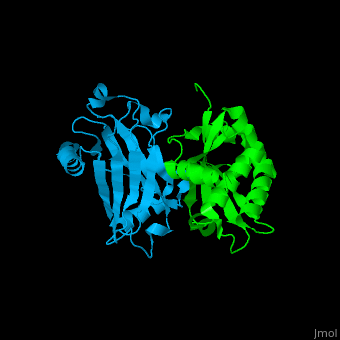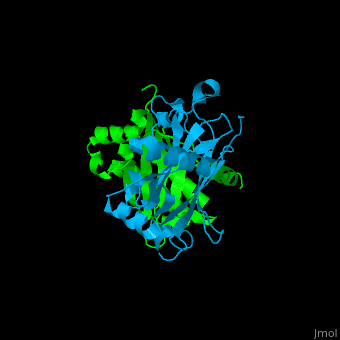
| - | <StructureSection load='1f3v' size=' | + | <StructureSection load='1f3v' size='450' side='right' caption='Human TRAF2 TRAF domain (green) complex with TNFR type 1 associated death domain protein TRADD (deepskyblue) (PDB code [[1d01]])' scene='70/708054/Cv/1'> |
== Function == | == Function == | ||
'''TNF receptor-associated factor''' (TRAF) are signal transducers and are involved in regulation of apoptosis, inflammation and antiviral response. There are 7 known TRAF proteins. All TRAF proteins share a C-terminal homology region named TRAF domain which can bind the cytoplasmic domain of receptors and other TRAF proteins<ref>PMID:11607847</ref>. '''TRAF1''' is the only TRAF which does not have N-terminal RING and zinc finger motifs. '''TRAF2, TRAF5 and TRAF6''' mediate activation of NF-κB and JNK. '''TRAF3''' mediates some innate immune receptor signals and regulates some post-translational modifications<ref>PMID:22017431</ref>. '''TRAF4''' is a binding partner of glycoproteins in platelets<ref>PMID:20946164</ref>. | '''TNF receptor-associated factor''' (TRAF) are signal transducers and are involved in regulation of apoptosis, inflammation and antiviral response. There are 7 known TRAF proteins. All TRAF proteins share a C-terminal homology region named TRAF domain which can bind the cytoplasmic domain of receptors and other TRAF proteins<ref>PMID:11607847</ref>. '''TRAF1''' is the only TRAF which does not have N-terminal RING and zinc finger motifs. '''TRAF2, TRAF5 and TRAF6''' mediate activation of NF-κB and JNK. '''TRAF3''' mediates some innate immune receptor signals and regulates some post-translational modifications<ref>PMID:22017431</ref>. '''TRAF4''' is a binding partner of glycoproteins in platelets<ref>PMID:20946164</ref>. | ||
Revision as of 09:21, 15 September 2016
| |||||||||||
3D Structures of TNF receptor-associated factor
Updated on 15-September-2016
References
- ↑ Bradley JR, Pober JS. Tumor necrosis factor receptor-associated factors (TRAFs). Oncogene. 2001 Oct 1;20(44):6482-91. PMID:11607847 doi:http://dx.doi.org/10.1038/sj.onc.1204788
- ↑ Hildebrand JM, Yi Z, Buchta CM, Poovassery J, Stunz LL, Bishop GA. Roles of tumor necrosis factor receptor associated factor 3 (TRAF3) and TRAF5 in immune cell functions. Immunol Rev. 2011 Nov;244(1):55-74. doi: 10.1111/j.1600-065X.2011.01055.x. PMID:22017431 doi:http://dx.doi.org/10.1111/j.1600-065X.2011.01055.x
- ↑ Arthur JF, Shen Y, Gardiner EE, Coleman L, Murphy D, Kenny D, Andrews RK, Berndt MC. TNF receptor-associated factor 4 (TRAF4) is a novel binding partner of glycoprotein Ib and glycoprotein VI in human platelets. J Thromb Haemost. 2011 Jan;9(1):163-72. doi: 10.1111/j.1538-7836.2010.04091.x. PMID:20946164 doi:http://dx.doi.org/10.1111/j.1538-7836.2010.04091.x
- ↑ Park YC, Ye H, Hsia C, Segal D, Rich RL, Liou HC, Myszka DG, Wu H. A novel mechanism of TRAF signaling revealed by structural and functional analyses of the TRADD-TRAF2 interaction. Cell. 2000 Jun 23;101(7):777-87. PMID:10892748