This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Aldehyde dehydrogenase
From Proteopedia
(Difference between revisions)
| Line 30: | Line 30: | ||
**[[3fx4]] – ALDH1 + NADP + inhibitor – pig<BR /> | **[[3fx4]] – ALDH1 + NADP + inhibitor – pig<BR /> | ||
| - | **[[1bxs]] – | + | **[[1bxs]] – sALDH1 + NAD - sheep<br /> |
| + | **[[5abm]] - sALDH1 + NAD derivative<br /> | ||
| + | **[[5ac0]] - sALDH1 + NAD + duocarmycin analog<br /> | ||
| + | **[[5ac1]], [[5ac2]] - sALDH1 + duocarmycin analog<br /> | ||
**[[1o9j]] – ALDH1 + NAD – Elephant shrew<br /> | **[[1o9j]] – ALDH1 + NAD – Elephant shrew<br /> | ||
**[[3rhj]], [[3rhl]], [[3rho]], [[3rhp]], [[3rhq]], [[3rhr]] – rALDH1 C- terminal (mutant) + NADP – rat<br /> | **[[3rhj]], [[3rhl]], [[3rho]], [[3rhp]], [[3rhq]], [[3rhr]] – rALDH1 C- terminal (mutant) + NADP – rat<br /> | ||
**[[3rhm]] - rALDH1 C- terminal<br /> | **[[3rhm]] - rALDH1 C- terminal<br /> | ||
| - | **[[1hso]] – hALDH1 α subunit + Zn + NAD<br /> | + | **[[1hso]] – hALDH1 α subunit + Zn + NAD - human<br /> |
| + | **[[4wj9]] - hALDH1 (mutant) <br /> | ||
| + | **[[4wpn]] - hALDH1 + inhibitor<br /> | ||
| + | **[[4wp7]] - hALDH1 (mutant) + inhibitor<br /> | ||
| + | **[[4wb9]] - hALDH1 + NAD derivative <br /> | ||
| + | **[[4x4l]] - hALDH1 + NAD derivative + inhibitor<br /> | ||
**[[1ht0]] – hALDH1 γ subunit (mutant) + Zn + NAD | **[[1ht0]] – hALDH1 γ subunit (mutant) + Zn + NAD | ||
*ALDH class 2 | *ALDH class 2 | ||
| - | **[[3n80]], [[1o05]], [[4kwf]] – hALDH2 residues 18-517 | + | **[[3n80]], [[1o05]], [[4kwf]] – hALDH2 residues 18-517<BR /> |
**[[3n81]], [[2onn]], [[2ono]], [[1zum]] – hALDH2 residues 18-517 (mutant) <BR /> | **[[3n81]], [[2onn]], [[2ono]], [[1zum]] – hALDH2 residues 18-517 (mutant) <BR /> | ||
**[[1ag8]] – bALDH NAD binding domain - bovine | **[[1ag8]] – bALDH NAD binding domain - bovine | ||
| Line 47: | Line 55: | ||
**[[3inj]], [[3inl]], [[4kwg]] - hALDH2 residues 18-517 + agonist<BR /> | **[[3inj]], [[3inl]], [[4kwg]] - hALDH2 residues 18-517 + agonist<BR /> | ||
**[[2vle]] - hALDH2 residues 24-517 + daidzin<BR /> | **[[2vle]] - hALDH2 residues 24-517 + daidzin<BR /> | ||
| - | **[[1nzx]], [[1nzz]], [[1o00]], [[1o02]], [[1cw3]] - hALDH2 residues 18-517 + NAD<BR /> | + | **[[1nzx]], [[1nzz]], [[1o00]], [[1o02]], [[1cw3]], [[4x2q]] - hALDH2 residues 18-517 + NAD<BR /> |
**[[3n82]], [[3n83]], [[2onm]], [[2onp]], [[1nzw]], [[1o04]] – hALDH2 residues 18-517 (mutant) + NADH<BR /> | **[[3n82]], [[3n83]], [[2onm]], [[2onp]], [[1nzw]], [[1o04]] – hALDH2 residues 18-517 (mutant) + NADH<BR /> | ||
**[[3sz9]] - hALDH2 residues 18-517 + inhibitor<br /> | **[[3sz9]] - hALDH2 residues 18-517 + inhibitor<br /> | ||
**[[1a4z]] – bALDH2 NAD-binding domain + NAD<BR /> | **[[1a4z]] – bALDH2 NAD-binding domain + NAD<BR /> | ||
| + | **[[1bi9]] – rALDH2 + NAD<br /> | ||
| + | **[[4pxl]] – ALDH2 + NAD - maize<br /> | ||
*ALDH2 ternary complex | *ALDH2 ternary complex | ||
| Line 73: | Line 83: | ||
**[[4mpb]], [[4nu9]], [[4q92]], [[4qto]] - Betaine-SaALDH – ''Staphylococcus aureus''<BR /> | **[[4mpb]], [[4nu9]], [[4q92]], [[4qto]] - Betaine-SaALDH – ''Staphylococcus aureus''<BR /> | ||
**[[4qje]] - Betaine-SaALDH (mutant)<br /> | **[[4qje]] - Betaine-SaALDH (mutant)<br /> | ||
| + | **[[4zxu]], [[4zwl]] - Betaine-SaALDH (mutant) + NAD<br /> | ||
**[[4cbb]] - Betaine-PaALDH - ''Pseudomonas aeruginosa''<br /> | **[[4cbb]] - Betaine-PaALDH - ''Pseudomonas aeruginosa''<br /> | ||
**[[1a4s]] - Betaine-cALDH - cod<BR /> | **[[1a4s]] - Betaine-cALDH - cod<BR /> | ||
| Line 83: | Line 94: | ||
**[[3r64]] – ALDH – ''Corynebacterium glutamicum''<br /> | **[[3r64]] – ALDH – ''Corynebacterium glutamicum''<br /> | ||
**[[3rhe]] – Benzaldehyde-ALDH – ''Legionella pneumophila''<br /> | **[[3rhe]] – Benzaldehyde-ALDH – ''Legionella pneumophila''<br /> | ||
| - | **[[3u4j]], [[4i3t]] – SmALDH – ''Sinorhizobium meliloti'' | + | **[[3u4j]], [[4i3t]] – SmALDH – ''Sinorhizobium meliloti''<br /> |
| + | **[[5dru]] - CpALDH – ''Clostridium phytofermentans''<br /> | ||
| + | **[[4qf6]], [[4ps9]] – BcALDH – ''Bacillus cereus''<br /> | ||
| + | **[[4qet]] – BcALDH (mutant)<br /> | ||
*NAD-dependent ALDH binary complexes | *NAD-dependent ALDH binary complexes | ||
| Line 89: | Line 103: | ||
**[[2w8q]] - Succinate–semi-hALDH family 5 + succinic acid<BR /> | **[[2w8q]] - Succinate–semi-hALDH family 5 + succinic acid<BR /> | ||
**[[4mpy]], [[4nea]] - Betaine-SaALDH + NAD<BR /> | **[[4mpy]], [[4nea]] - Betaine-SaALDH + NAD<BR /> | ||
| - | **[[4qn2]] - Betaine-SaALDH (mutant) + NAD<br /> | + | **[[4qn2]], [[5ez4]], [[5eyu]], [[5dib]] - Betaine-SaALDH (mutant) + NAD<br /> |
**[[1bpw]] - Betaine-cALDH + NAD<BR /> | **[[1bpw]] - Betaine-cALDH + NAD<BR /> | ||
**[[2j6l]] – Aminoadipate-semi-hALDH family 7 + NAD<BR /> | **[[2j6l]] – Aminoadipate-semi-hALDH family 7 + NAD<BR /> | ||
**[[2cfi]] - Formyltetrahydrofolate-hALDH hydrolase domain + formyltetrahydropterin<BR /> | **[[2cfi]] - Formyltetrahydrofolate-hALDH hydrolase domain + formyltetrahydropterin<BR /> | ||
| - | **[[2nad]] - Formate-PaALDH | + | **[[2nad]] - Formate-PaALDH + NAD <BR /> |
| - | + | ||
**[[3l4p]], [[1sij]] – ALDH + AsO3 – ''Desulfovibrio gigas''<br /> | **[[3l4p]], [[1sij]] – ALDH + AsO3 – ''Desulfovibrio gigas''<br /> | ||
| - | **[[4a0m]] – spALDH + NAD – spinach<br /> | + | **[[4a0m]], [[4v37]] – spALDH + NAD – spinach<br /> |
| + | **[[5a2d]], [[4v3f]] – spALDH + choline<br /> | ||
**[[3iwk]] – amino-ALDH + NAD – pea<br /> | **[[3iwk]] – amino-ALDH + NAD – pea<br /> | ||
**[[3ju8]] - Succinylglutamic–semi-PaALDH + NAD <br /> | **[[3ju8]] - Succinylglutamic–semi-PaALDH + NAD <br /> | ||
**[[4caz]] - Betaine-PaALDH + NAD derivative<br /> | **[[4caz]] - Betaine-PaALDH + NAD derivative<br /> | ||
| - | **[[4c3s]] - | + | **[[4c3s]] - CpALDH + NAD <br /> |
| + | **[[5dbv]] - CpALDH + CoA <br /> | ||
**[[4i3u]] – SmALDH + phosphonoacetaldehyde<br /> | **[[4i3u]] – SmALDH + phosphonoacetaldehyde<br /> | ||
| + | **[[4pt0]] – BcALDH + NAD<br /> | ||
*NAD-dependent ALDH ternary complexes | *NAD-dependent ALDH ternary complexes | ||
| Line 119: | Line 135: | ||
**[[3ifg]], [[3ifh]] - Succinate–semi-ALDH – ''Burkholderia pseudomallei''<br /> | **[[3ifg]], [[3ifh]] - Succinate–semi-ALDH – ''Burkholderia pseudomallei''<br /> | ||
**[[3rh9]] - Succinate–semi-ALDH – ''Marinobacter aquaeolei''<br /> | **[[3rh9]] - Succinate–semi-ALDH – ''Marinobacter aquaeolei''<br /> | ||
| - | **[[3uw3]] - aspartate–semi- | + | **[[3uw3]] - aspartate–semi-BtALDH – ''Burkholderia thailandensis''<br /> |
**[[3v4c]] - SmALDH<br /> | **[[3v4c]] - SmALDH<br /> | ||
**[[4gac]] – ALDH – mouse<br /> | **[[4gac]] – ALDH – mouse<br /> | ||
| + | **[[5eeb]] - PfALDH – ''Pyrobaculum ferrireducens''<br /> | ||
*NADP-dependent ALDH binary complexes | *NADP-dependent ALDH binary complexes | ||
| Line 134: | Line 151: | ||
**[[3jz4]] - Succinate–semi-EcALDH + NADP<br /> | **[[3jz4]] - Succinate–semi-EcALDH + NADP<br /> | ||
**[[3hsk]] - aspartate–semi-ALDH + NADP – ''Candida albicans''<br /> | **[[3hsk]] - aspartate–semi-ALDH + NADP – ''Candida albicans''<br /> | ||
| - | **[[4h73]], [[4nmj]], [[4nmk]] - ALDH + NADP – '' | + | **[[4h73]], [[4nmj]], [[4nmk]], [[5f2c]], [[5exf]], [[5euy]], [[5ekc]], [[5ek6]] - PfALDH + NADP <br /> |
| + | **[[5kf0]] - ALDH + NADP – ''Burkholderia vietnamiensis''<br /> | ||
| + | **[[5jry]] - ALDH + NADP – ''Burkholderia multivorans''<br /> | ||
| + | **[[5j6b]] - BtALDH + NADP <br /> | ||
| + | **[[4pt3]] – BcALDH + NADP<br /> | ||
*NADP-dependent ALDH ternary complexes | *NADP-dependent ALDH ternary complexes | ||
Revision as of 10:34, 27 February 2017
| |||||||||||
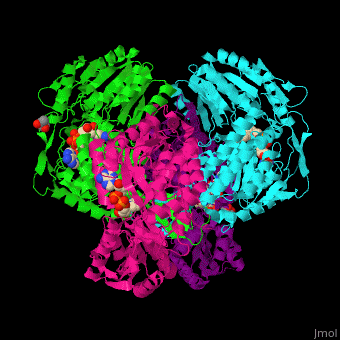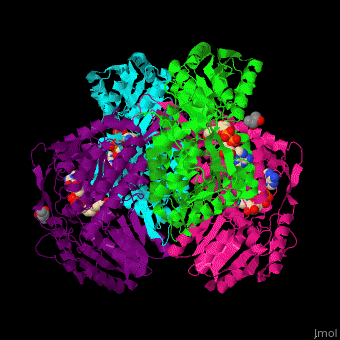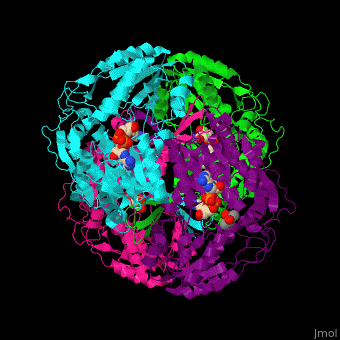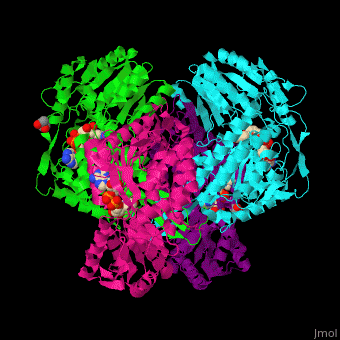
3D Structures of Aldehyde dehydrogenase
Updated on 27-February-2017
References
- ↑ Keller, Markus A.; Zander, Ulrich; Fuchs, Julian E.; Kreutz, Christoph; Watschinger, Katrin et al. (2014). A gatekeeper helix determines the substrate specificity of Sjögren–Larsson Syndrome enzyme fatty aldehyde dehydrogenase. Nature Communications vol. 5.