This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
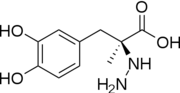
Carbidopa
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
== Function == | == Function == | ||
| - | <StructureSection load='1js3'=size='340' side='right' caption='Crystal structure of DOPA Decarboxylase in complex with the inhibitor carbidopa PDB:[[1js3]]' scene=''><scene name='pdbligand=142:CARBIDOPA'>Carbidopa</scene> (Lodosyn) is a drug given, in conjunction with Levadopa, to those with Parkinson's Disease in order to inhibit the extracranial metabolism of Levadopa to dopamine, allowing a greater portion of the drug to cross the blood-brain barrier and produce the desired anti-parkinsonian effects. It works by inhibiting the enzymatic activity of aromatic-L-amino-acid decarboxylase ([[DOPA Decarboxylase]] or DDC)<ref name="one">PMID:11106255</ref> | + | <StructureSection load='1js3'=size='340' side='right' caption='Crystal structure of DOPA Decarboxylase in complex with the inhibitor carbidopa PDB:[[1js3]]' scene=''><scene name='pdbligand=142:CARBIDOPA'>Carbidopa</scene> (Lodosyn) is a drug given, in conjunction with Levadopa, to those with Parkinson's Disease in order to inhibit the extracranial metabolism of Levadopa to dopamine, allowing a greater portion of the drug to cross the blood-brain barrier and produce the desired anti-parkinsonian effects in the nerve cell. It works by inhibiting the enzymatic activity of aromatic-L-amino-acid decarboxylase ([[DOPA Decarboxylase]] or DDC)<ref name="one">PMID:11106255</ref>. |
== Structure == | == Structure == | ||
| Line 6: | Line 6: | ||
== Molecular Mechanism == | == Molecular Mechanism == | ||
| - | The conversion of L-DOPA into dopamine is catalyzed by the vitamin B6 (<scene name='pdbligand=PLP:PYRIDOXAL-5-PHOSPHATE'>PLP</scene>)-dependent enzyme DDC, an enzyme abundant in the nervous system as well as kidneys of humans<ref name="three">PMID: 7651438</ref> The catalytically active form of DDC is a homodimer, a feature typical of this class of enzymes<ref name= "four">PMID: 10673430</ref>. DDCs active site is located between the two monomers but is | + | The conversion of L-DOPA into dopamine is catalyzed by the vitamin B6 (<scene name='pdbligand=PLP:PYRIDOXAL-5-PHOSPHATE'>PLP</scene>)-dependent enzyme DDC, an enzyme abundant in the nervous system as well as kidneys of humans<ref name="three">PMID: 7651438</ref> The catalytically active form of DDC is a homodimer, a feature typical of this class of enzymes<ref name= "four">PMID: 10673430</ref>. DDCs active site is located between the two monomers but is mainly composed of residues from only one of the monomers. The cofactor PLP binds to Lys 303 through a Schiff base linkage and a salt bridge between the carboxylate group of Asp 271 and the protonated pyridine nitrogen of PLP, which acts as a strong electron sink capable of stabilizing the carbanionic intermediates produced by active DDC. the cofactor is further stabilized in the enzyme through a network of hydrogen bonds. Carbidopa works by forming a hydrazone linkage with the PLP cofactor through its hydrazine moiety and blocking the DDC <scene name='74/746001/Active_site_dopa/4'>active site</scene> residues Ile 101' and Phe 103' in the substrate binding pocket with its catechol ring<ref name="five">doi:10.1038/nsb1101-963</ref>. Due to the fact that Carbidopa cannot cross the blood-brain barrier, its inhibiting effects only are displayed in the periphery. |
== Disease in Humans == | == Disease in Humans == | ||
Revision as of 17:43, 16 November 2016
Function
| |||||||||||
References
- ↑ Gilbert JA, Frederick LM, Ames MM. The aromatic-L-amino acid decarboxylase inhibitor carbidopa is selectively cytotoxic to human pulmonary carcinoid and small cell lung carcinoma cells. Clin Cancer Res. 2000 Nov;6(11):4365-72. PMID:11106255
- ↑ https://pubchem.ncbi.nlm.nih.gov/compound/carbidopa#section=Top
- ↑ Opacka-Juffry J, Brooks DJ. L-dihydroxyphenylalanine and its decarboxylase: new ideas on their neuroregulatory roles. Mov Disord. 1995 May;10(3):241-9. PMID:7651438 doi:http://dx.doi.org/10.1002/mds.870100302
- ↑ Schneider G, Kack H, Lindqvist Y. The manifold of vitamin B6 dependent enzymes. Structure. 2000 Jan 15;8(1):R1-6. PMID:10673430
- ↑ Burkhard P, Dominici P, Borri-Voltattorni C, Jansonius JN, Malashkevich VN. Structural insight into Parkinson's disease treatment from drug-inhibited DOPA decarboxylase. Nat Struct Biol. 2001 Nov;8(11):963-7. PMID:11685243 doi:http://dx.doi.org/10.1038/nsb1101-963
- ↑ Feany MB, Bender WW. A Drosophila model of Parkinson's disease. Nature. 2000 Mar 23;404(6776):394-8. PMID:10746727 doi:http://dx.doi.org/10.1038/35006074