This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
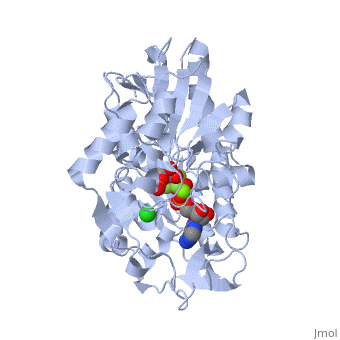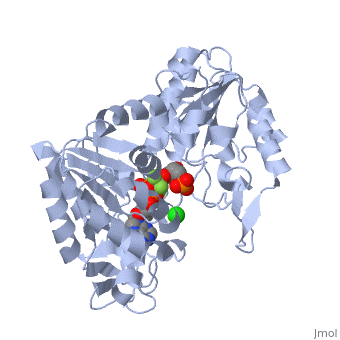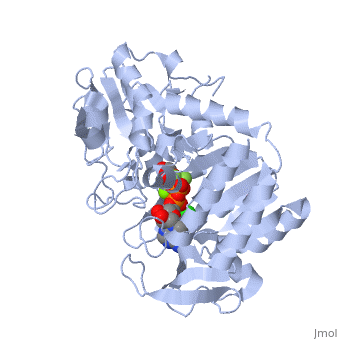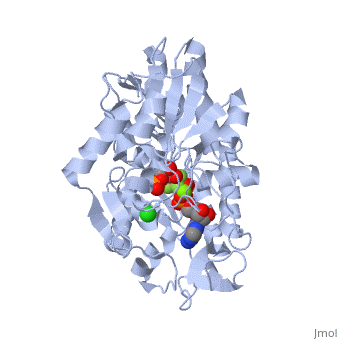
Phosphoglycerate Kinase
From Proteopedia
(Difference between revisions)
| Line 57: | Line 57: | ||
**[[4dg5]] – PGK – ''Staphylococcus aureus''<br /> | **[[4dg5]] – PGK – ''Staphylococcus aureus''<br /> | ||
**[[4ehj]] – FtPGK – ''Francisella tularensis''<br /> | **[[4ehj]] – FtPGK – ''Francisella tularensis''<br /> | ||
| + | **[[5bt8]] – PGK – ''Acinetobacter baumannii''<br /> | ||
*Phosphoglycerate kinase binary complex | *Phosphoglycerate kinase binary complex | ||
| Line 69: | Line 70: | ||
**[[2cun]] – PGK + PGA – ''Pyrococcus horikoshii''<BR /> | **[[2cun]] – PGK + PGA – ''Pyrococcus horikoshii''<BR /> | ||
**[[2p9t]] – mPGK2 + PGA<BR /> | **[[2p9t]] – mPGK2 + PGA<BR /> | ||
| - | **[[2xe6]], [[3c39]] – hPGK1 + PGA<BR /> | + | **[[2xe6]], [[3c39]] – hPGK1 + PGA - human<BR /> |
**[[2zgv]] - hPGK1 + ADP<BR /> | **[[2zgv]] - hPGK1 + ADP<BR /> | ||
**[[3c3b]], [[3c3c]] - hPGK1 + CDP <br /> | **[[3c3b]], [[3c3c]] - hPGK1 + CDP <br /> | ||
**[[3zoz]] – hPGK1 + Br<br /> | **[[3zoz]] – hPGK1 + Br<br /> | ||
**[[3zlb]] - PGK + ANP – ''Streptococcus pneumoniae''<br /> | **[[3zlb]] - PGK + ANP – ''Streptococcus pneumoniae''<br /> | ||
| + | **[[4ng4]] – PGK + ADP – ''Coxiella burnetii''<br /> | ||
*Phosphoglycerate kinase ternary complex | *Phosphoglycerate kinase ternary complex | ||
| Line 83: | Line 85: | ||
**[[3pgk]] - yPGK + ATP + PGA<BR /> | **[[3pgk]] - yPGK + ATP + PGA<BR /> | ||
**[[13pk]] - TbPGK + ADP + PGA<BR /> | **[[13pk]] - TbPGK + ADP + PGA<BR /> | ||
| - | **[[2y3i]], [[2ybe]], [[2xe7]], [[2x13]], [[3c3a]] – hPGK1 + ADP + PGA | + | **[[2y3i]], [[2ybe]], [[2xe7]], [[2x13]], [[3c3a]] – hPGK1 + ADP + PGA <BR /> |
| + | **[[2x15]] - hPGK1 + bisphosphoglycerate + ADP<br /> | ||
**[[2xe8]] - hPGK1 + AMPPNP + PGA<BR /> | **[[2xe8]] - hPGK1 + AMPPNP + PGA<BR /> | ||
**[[2x14]] - hPGK1 (mutant) + AMPPCP + PGA<BR /> | **[[2x14]] - hPGK1 (mutant) + AMPPCP + PGA<BR /> | ||
Revision as of 07:38, 5 September 2017
| |||||||||||
3D structures of phosphoglycerate kinase
Updated on 05-September-2017
Additional Resources
For additional information, see: Carbohydrate Metabolism
References
- ↑ Auerbach, Gunter et al. 1997. Closed Structure of phosphoglycerate kinase from Thermotoga maritima reveals the catalytic mechanism and determinants of thermal stability. Structure. 5:1475-1483.
- ↑ Lallemand P, Chaloin L, Roy B, Barman T, Bowler MW, Lionne C. Interaction of human 3-phosphoglycerate kinase with its two substrates: is substrate antagonism a kinetic advantage? J Mol Biol. 2011 Jun 24;409(5):742-57. Epub 2011 Apr 27. PMID:21549713 doi:10.1016/j.jmb.2011.04.048
- ↑ Voet, Donald et al. 2008. Fundamentals of Biochemistry. 3rd ed. 499
- ↑ Auerbach, Gunter et al. 1997. Closed Structure of phosphoglycerate kinase from Thermotoga maritima reveals the catalytic mechanism and determinants of thermal stability. Structure. 5:1475-1483.
- ↑ Blake and Rice. 1981. Phosphoglycerate kinase. Philosophical Transactions of the Royal Society of London. 293:93-104.
- ↑ Vas, M, Varga, A et al. 2010. Insight into the Mechanism of of Domain Movements and their Role in Enzyme Function: Example of 3-Phosphoglycerate kinase. Current Protein and Peptide Science. Jan 21, 2010. (Epub ahead of publication).
- ↑ Harnan, G. et al. 1992. Domain Motions in Phosphoglycerate Kinase: Determination of Interdomain Distance Distribution by Site Specific Labeling and Time Resolved Flourescense Energy Transfer. PNAS. 89:11764-11768.
- ↑ Auerbach, Gunter et al. 1997. Closed Structure of phosphoglycerate kinase from Thermotoga maritima reveals the catalytic mechanism and determinants of thermal stability. Structure. 5:1475-1483.
- ↑ Auerbach, Gunter et al. 1997. Closed Structure of phosphoglycerate kinase from Thermotoga maritima reveals the catalytic mechanism and determinants of thermal stability. Structure. 5:1475-1483.
- ↑ Scopes, Robert. 1977. The Steady State Kinetics of Yeast Phosphoglycerate Kinase. European Journal of Biochemistry. 85, 503-516
- ↑ Macioszek, Jerzy et al. 1990. Kinetics of the Two-Enzyme Phosphoglycerate Kinase/Glyceraldehyde-3-Phosphate Dehydrogenase Couple. Plant Physiology 94: 291-296.
- ↑ Shaobo, Wu et al. 2009. PGK1 expression responds to freezing, anoxia, and dehydration stresses in freeze tolerant wood frog, Rana sylvatica. Journal of Experimental Zoology. 311, 57-67
- ↑ Hogg, PJ. 2002. Biological Regulation through protein disulfide bond cleavage. Redox Report. 7(2), 71-77.
Proteopedia Page Contributors and Editors (what is this?)
Shane Harmon, Michal Harel, Joel L. Sussman, Brandon Tritle, David Canner, Alexander Berchansky