This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Vitamin D receptor
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
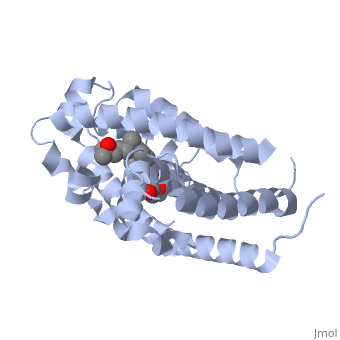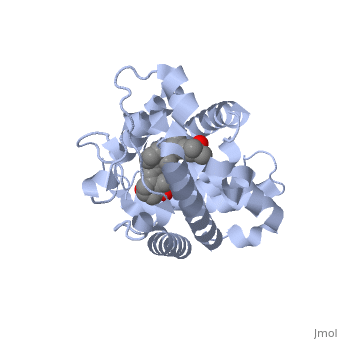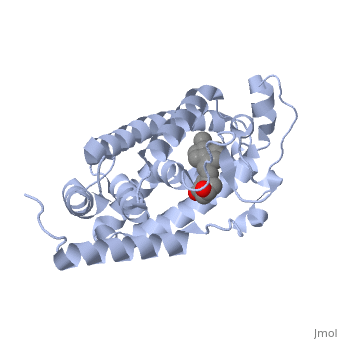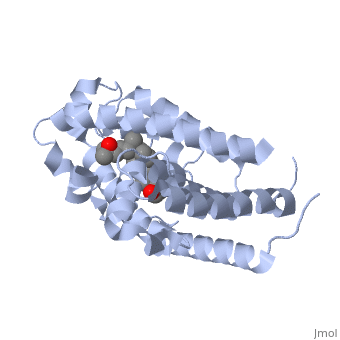
<StructureSection load='1db1' size='350' side='right' caption='Structure of human vitamin D receptor ligand-binding domain complex with vitamin D (PDB entry [[1db1]])' scene=''> | <StructureSection load='1db1' size='350' side='right' caption='Structure of human vitamin D receptor ligand-binding domain complex with vitamin D (PDB entry [[1db1]])' scene=''> | ||
== Introduction == | == Introduction == | ||
| - | '''Vitamin D receptor''' (<scene name='56/562378/Vit_d_receptor_3m7r/3'>VDR</scene>) is a transcription factor. Upon binding to vitamin D, VDR forms a heterodimer with retinoid-X receptor and binds to hormone response receptors on DNA causing gene expression. The <scene name='56/562378/Vit_d_receptor_ligand/1'>vitamin D hormone</scene> (in green) binds to receptors in its target cells, controlling the synthesis of many different proteins involved in calcium transport and utilization. VDR contains two domains: a <scene name='56/562378/Lbd/1'>ligand binding domain (LBD)</scene> that binds to the hormone (grey) and <scene name='56/562378/Dbd/2'>DNA-binding domain (DBD)</scene> that binds to DNA. (Green and blue are two same VDR structures). It pairs up with a similar protein, 9-cis retinoic acid receptor (RXR), and together they bind to the DNA, activating synthesis in some cases and repressing it in others. | + | '''Vitamin D receptor''' (<scene name='56/562378/Vit_d_receptor_3m7r/3'>VDR</scene>) is a transcription factor. Upon binding to vitamin D, VDR forms a heterodimer with retinoid-X receptor and binds to hormone response receptors on DNA causing gene expression. The <scene name='56/562378/Vit_d_receptor_ligand/1'>vitamin D hormone</scene> (in green) binds to receptors in its target cells, controlling the synthesis of many different proteins involved in calcium transport and utilization. VDR contains two domains: a <scene name='56/562378/Lbd/1'>ligand binding domain (LBD)</scene> that binds to the hormone (grey) and <scene name='56/562378/Dbd/2'>DNA-binding domain (DBD)</scene> that binds to DNA. (Green and blue are two same VDR structures). It pairs up with a similar protein, 9-cis retinoic acid receptor (RXR), and together they bind to the DNA, activating synthesis in some cases and repressing it in others<ref>PMID:11425573</ref>. |
<!-- | <!-- | ||
| Line 71: | Line 71: | ||
**[[1kb6]] – hVDR DBD + osteocalcin response element DNA<br /> | **[[1kb6]] – hVDR DBD + osteocalcin response element DNA<br /> | ||
}} | }} | ||
| - | + | ==References== | |
| + | <references /> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Revision as of 11:04, 13 December 2016
| |||||||||||
3D structures of vitamin D receptor
Updated on 13-December-2016
References
- ↑ Choi M, Yamamoto K, Masuno H, Nakashima K, Taga T, Yamada S. Ligand recognition by the vitamin D receptor. Bioorg Med Chem. 2001 Jul;9(7):1721-30. PMID:11425573
- ↑ Hughes MR, Malloy PJ, Kieback DG, Kesterson RA, Pike JW, Feldman D, O'Malley BW. Point mutations in the human vitamin D receptor gene associated with hypocalcemic rickets. Science. 1988 Dec 23;242(4886):1702-5. PMID:2849209
- ↑ Yagi H, Ozono K, Miyake H, Nagashima K, Kuroume T, Pike JW. A new point mutation in the deoxyribonucleic acid-binding domain of the vitamin D receptor in a kindred with hereditary 1,25-dihydroxyvitamin D-resistant rickets. J Clin Endocrinol Metab. 1993 Feb;76(2):509-12. PMID:8381803
- ↑ Saijo T, Ito M, Takeda E, Huq AH, Naito E, Yokota I, Sone T, Pike JW, Kuroda Y. A unique mutation in the vitamin D receptor gene in three Japanese patients with vitamin D-dependent rickets type II: utility of single-strand conformation polymorphism analysis for heterozygous carrier detection. Am J Hum Genet. 1991 Sep;49(3):668-73. PMID:1652893
- ↑ Sone T, Marx SJ, Liberman UA, Pike JW. A unique point mutation in the human vitamin D receptor chromosomal gene confers hereditary resistance to 1,25-dihydroxyvitamin D3. Mol Endocrinol. 1990 Apr;4(4):623-31. PMID:2177843
- ↑ Malloy PJ, Weisman Y, Feldman D. Hereditary 1 alpha,25-dihydroxyvitamin D-resistant rickets resulting from a mutation in the vitamin D receptor deoxyribonucleic acid-binding domain. J Clin Endocrinol Metab. 1994 Feb;78(2):313-6. PMID:8106618
- ↑ Kristjansson K, Rut AR, Hewison M, O'Riordan JL, Hughes MR. Two mutations in the hormone binding domain of the vitamin D receptor cause tissue resistance to 1,25 dihydroxyvitamin D3. J Clin Invest. 1993 Jul;92(1):12-6. PMID:8392085 doi:http://dx.doi.org/10.1172/JCI116539
- ↑ Rut AR, Hewison M, Kristjansson K, Luisi B, Hughes MR, O'Riordan JL. Two mutations causing vitamin D resistant rickets: modelling on the basis of steroid hormone receptor DNA-binding domain crystal structures. Clin Endocrinol (Oxf). 1994 Nov;41(5):581-90. PMID:7828346
- ↑ Lin NU, Malloy PJ, Sakati N, al-Ashwal A, Feldman D. A novel mutation in the deoxyribonucleic acid-binding domain of the vitamin D receptor causes hereditary 1,25-dihydroxyvitamin D-resistant rickets. J Clin Endocrinol Metab. 1996 Jul;81(7):2564-9. PMID:8675579
- ↑ Whitfield GK, Selznick SH, Haussler CA, Hsieh JC, Galligan MA, Jurutka PW, Thompson PD, Lee SM, Zerwekh JE, Haussler MR. Vitamin D receptors from patients with resistance to 1,25-dihydroxyvitamin D3: point mutations confer reduced transactivation in response to ligand and impaired interaction with the retinoid X receptor heterodimeric partner. Mol Endocrinol. 1996 Dec;10(12):1617-31. PMID:8961271
- ↑ Malloy PJ, Eccleshall TR, Gross C, Van Maldergem L, Bouillon R, Feldman D. Hereditary vitamin D resistant rickets caused by a novel mutation in the vitamin D receptor that results in decreased affinity for hormone and cellular hyporesponsiveness. J Clin Invest. 1997 Jan 15;99(2):297-304. PMID:9005998 doi:10.1172/JCI119158
- ↑ Fujiki R, Kim MS, Sasaki Y, Yoshimura K, Kitagawa H, Kato S. Ligand-induced transrepression by VDR through association of WSTF with acetylated histones. EMBO J. 2005 Nov 16;24(22):3881-94. Epub 2005 Oct 27. PMID:16252006 doi:10.1038/sj.emboj.7600853
- ↑ Rochel N, Wurtz JM, Mitschler A, Klaholz B, Moras D. The crystal structure of the nuclear receptor for vitamin D bound to its natural ligand. Mol Cell. 2000 Jan;5(1):173-9. PMID:10678179
- ↑ Eelen G, Verlinden L, Rochel N, Claessens F, De Clercq P, Vandewalle M, Tocchini-Valentini G, Moras D, Bouillon R, Verstuyf A. Superagonistic action of 14-epi-analogs of 1,25-dihydroxyvitamin D explained by vitamin D receptor-coactivator interaction. Mol Pharmacol. 2005 May;67(5):1566-73. Epub 2005 Feb 22. PMID:15728261 doi:10.1124/mol.104.008730
- ↑ Hourai S, Fujishima T, Kittaka A, Suhara Y, Takayama H, Rochel N, Moras D. Probing a water channel near the A-ring of receptor-bound 1 alpha,25-dihydroxyvitamin D3 with selected 2 alpha-substituted analogues. J Med Chem. 2006 Aug 24;49(17):5199-205. PMID:16913708 doi:http://dx.doi.org/10.1021/jm0604070
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Jaime Prilusky, Isita Amin