This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1069
From Proteopedia
| Line 39: | Line 39: | ||
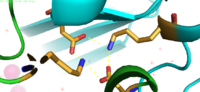
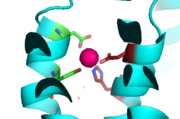
<scene name='69/694236/Site_c/1'>Binding Site C</scene> has vastly opposite properties from what is seen in binding site A. It is located on the TM2-TM3 loop on the cytoplasmic membrane and between the two C-terminus domain interfaces. Here, there is a binuclear coordination of zinc between the <scene name='69/694236/Asp285/3'>Asp285</scene> residue that bridges the zinc ions together and the four coordinating residues (His232, His248, His283 and His261). The Asp285 residue is conserved, meaning it does not have outer shell constraints. However, the four histidine residues all have outer shell constraints. These constraints consist of hydrogen bonds to the residues surrounding the binding site. These hydrogen bonds can form bidentate bonds, which means that the hydrogen bond attaches to a metal in two places. These bonds in turn create an extensive network of interactions at the CTD interface, and it is these interactions that allow for stability and strengthening of the CTD-CTD association. | <scene name='69/694236/Site_c/1'>Binding Site C</scene> has vastly opposite properties from what is seen in binding site A. It is located on the TM2-TM3 loop on the cytoplasmic membrane and between the two C-terminus domain interfaces. Here, there is a binuclear coordination of zinc between the <scene name='69/694236/Asp285/3'>Asp285</scene> residue that bridges the zinc ions together and the four coordinating residues (His232, His248, His283 and His261). The Asp285 residue is conserved, meaning it does not have outer shell constraints. However, the four histidine residues all have outer shell constraints. These constraints consist of hydrogen bonds to the residues surrounding the binding site. These hydrogen bonds can form bidentate bonds, which means that the hydrogen bond attaches to a metal in two places. These bonds in turn create an extensive network of interactions at the CTD interface, and it is these interactions that allow for stability and strengthening of the CTD-CTD association. | ||
| - | + | ==Zn<sup>2+</sup> Induced Conformation Change== | |
Conformation changes occur in the TMD and CTD, both of which are heavily influenced by the presence of Zn<sup>2+</sup>. The conformation change directly involved with Zn<sup>2+</sup>/H<sup>+</sup> antiport occurs in the TMD as helix pivoting controls what environment site A is available to. Conformation change occurs when the transmembrane helix pairs TM3-TM6 pivot around cation binding site. It is believed that the energy for TMD conformation change comes from energy of binding each substrate. Changing to the outward from the inward-facing conformation causes a shift in TM5 which disrupts the tetrahedral geometry of active site A. This in turn decreases binding affinity site A has for Zn<sup>2+</sup> and causes Zn<sup>2+</sup> to leave site A which then favors change back to inward-facing conformation. | Conformation changes occur in the TMD and CTD, both of which are heavily influenced by the presence of Zn<sup>2+</sup>. The conformation change directly involved with Zn<sup>2+</sup>/H<sup>+</sup> antiport occurs in the TMD as helix pivoting controls what environment site A is available to. Conformation change occurs when the transmembrane helix pairs TM3-TM6 pivot around cation binding site. It is believed that the energy for TMD conformation change comes from energy of binding each substrate. Changing to the outward from the inward-facing conformation causes a shift in TM5 which disrupts the tetrahedral geometry of active site A. This in turn decreases binding affinity site A has for Zn<sup>2+</sup> and causes Zn<sup>2+</sup> to leave site A which then favors change back to inward-facing conformation. | ||
Revision as of 21:07, 29 March 2017
Introduction
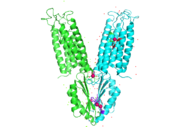
Zinc transporter YiiP is an integral membrane protein found in the membrane of Esherichia coli. YiiP is a member of the cation diffusion facilitator family. Members of this family occur all throughout the biological realm. These diffusion facilitators export divalent transition metal ions from the cytoplasm to the extracellular space [1]. They work to regulate the amount of divalent metals inside of the cell, which is biologically relevant because while these metals are necessary for different biological functions, they can prove fatal to the cell in excess amounts. Zinc is essential for the growth and development of cells and zinc levels can affect everything from gene expression to immune response. While YiiP is an integral membrane protein in the cells of Escherichia coli, understanding the mechanism of regulation behind it can help researcher's better understand the cation diffusion facilitator equivalents in eukaryotic cells.
| |||||||||||