This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1063
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
==='''Structural Overview'''=== | ==='''Structural Overview'''=== | ||
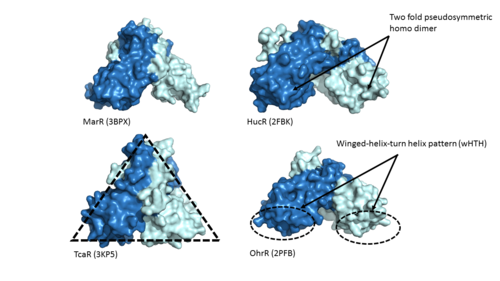
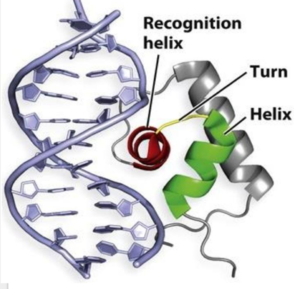
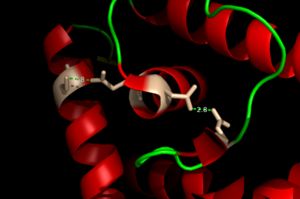
| - | One of the two functional domains of AdcR is <scene name='69/694230/Dimerization_domain/3'> dimerization domain</scene>. This domain connects and stabilizes the two pseudosymmetric dimers and is composed of the <scene name='69/694230/Alpha_1/1'>α1 helix</scene>, the C-terminus of the <scene name='69/694230/Alpha_five/1'>α5 helix</scene> , and the <scene name='69/694230/Alpha_6/1'>α6 helix</scene>. This domain is connected to the DNA binding domain by the long α5 helix. The DNA binding domain stabilizes the major and minor groove of DNA via the winged helix-turn-helix motif. Binding of Zinc allows AdcR to bind DNA and activate the transcription of high-affinity Zinc specific uptake transporters. The binding of Zinc induces a conformational change that allows for a hydrogen bond network between helices of the binding domain. It is believed that this hydrogen bond network is the allosteric activator needed to expose residues that bind the bases along the major groove of the DNA <ref name="guerra">PMID:22085181</ref>. The binding sites are found adjacent to the DNA binding domain. | + | One of the two functional domains of AdcR is <scene name='69/694230/Dimerization_domain/3'> dimerization domain</scene>. This domain connects and stabilizes the two pseudosymmetric dimers and is composed of the <scene name='69/694230/Alpha_1/1'>α1 helix</scene>, the C-terminus of the <scene name='69/694230/Alpha_five/1'>α5 helix</scene> , and the <scene name='69/694230/Alpha_6/1'>α6 helix</scene>. This domain is connected to the DNA binding domain by the long α5 helix. The DNA binding domain stabilizes the major and minor groove of DNA via the winged helix-turn-helix motif. Binding of Zinc allows AdcR to bind DNA and activate the transcription of high-affinity Zinc specific uptake transporters. The binding of Zinc to the <scene name='69/694230/2_binding_sites/2'>Zinc binding pocket</scene> induces a conformational change that allows for a hydrogen bond network between helices of the binding domain. It is believed that this hydrogen bond network is the allosteric activator needed to expose residues that bind the bases along the major groove of the DNA <ref name="guerra">PMID:22085181</ref>. The binding sites are found adjacent to the DNA binding domain. |
Revision as of 17:58, 21 April 2017
Adhesin Competence Regulator
| |||||||||||
References
- ↑ Sanson M, Makthal N, Flores AR, Olsen RJ, Musser JM, Kumaraswami M. Adhesin competence repressor (AdcR) from Streptococcus pyogenes controls adaptive responses to zinc limitation and contributes to virulence. Nucleic Acids Res. 2015 Jan;43(1):418-32. doi: 10.1093/nar/gku1304. Epub 2014 Dec, 15. PMID:25510500 doi:http://dx.doi.org/10.1093/nar/gku1304
- ↑ Fraústo da Silva J, Williams R. The Biological Chemistry of Elements: The Inorganic Chemistry of Life. Second ed. Oxford University Press; Oxford: 2001.
- ↑ Ma Z, Jacobsen FE, Giedroc DP. Coordination chemistry of bacterial metal transport and sensing. Chem Rev. 2009 Oct;109(10):4644-81. doi: 10.1021/cr900077w. PMID:19788177 doi:http://dx.doi.org/10.1021/cr900077w
- ↑ 4.0 4.1 4.2 4.3 Guerra AJ, Dann CE, Giedroc DP. Crystal Structure of the Zinc-Dependent MarR Family Transcriptional Regulator AdcR in the Zn(II)-Bound State. J Am Chem Soc. 2011 Nov 21. PMID:22085181 doi:10.1021/ja2080532
- ↑ 5.0 5.1 Reyes-Caballero H, Guerra AJ, Jacobsen FE, Kazmierczak KM, Cowart D, Koppolu UM, Scott RA, Winkler ME, Giedroc DP. The metalloregulatory zinc site in Streptococcus pneumoniae AdcR, a zinc-activated MarR family repressor. J Mol Biol. 2010 Oct 22;403(2):197-216. doi: 10.1016/j.jmb.2010.08.030. Epub 2010, Sep 8. PMID:20804771 doi:http://dx.doi.org/10.1016/j.jmb.2010.08.030