This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sucrase-isomaltase
From Proteopedia
(Difference between revisions)
| Line 7: | Line 7: | ||
Some other key enzymes that capitalize on this characteristic in the small intestine are: glucoamylase (Maltase), lactase, and peptidases. Among these common enzymes however is SI, which is important because it is critically involved in the final stage of carbohydrate digestion. <ref> http://www.uniprot.org/uniprot/P14410</ref> Specifically, this enzyme works by catalyzing the conversion of isomaltose into glucose molecules. Cleaving bonds with water (hydrolysis), SI interacts with glucosidic linkages produced by alpha-amylase. <ref>Maureen Barlow Pugh, ed. (2000). Stedman's Medical Dictionary (27th ed.). Baltimore, Maryland, USA: Lippincott Williams & Wilkins. p. 65. ISBN 978-0-683-40007-6. </ref> | Some other key enzymes that capitalize on this characteristic in the small intestine are: glucoamylase (Maltase), lactase, and peptidases. Among these common enzymes however is SI, which is important because it is critically involved in the final stage of carbohydrate digestion. <ref> http://www.uniprot.org/uniprot/P14410</ref> Specifically, this enzyme works by catalyzing the conversion of isomaltose into glucose molecules. Cleaving bonds with water (hydrolysis), SI interacts with glucosidic linkages produced by alpha-amylase. <ref>Maureen Barlow Pugh, ed. (2000). Stedman's Medical Dictionary (27th ed.). Baltimore, Maryland, USA: Lippincott Williams & Wilkins. p. 65. ISBN 978-0-683-40007-6. </ref> | ||
| - | |||
== Structure == | == Structure == | ||
| Line 45: | Line 44: | ||
-The gene on which SI processes occur is located on chromosome 3 and is composed of 48 exons. | -The gene on which SI processes occur is located on chromosome 3 and is composed of 48 exons. | ||
</StructureSection> | </StructureSection> | ||
| - | ==3D structures of surase-isomaltase== | ||
| - | |||
| - | Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | ||
| - | |||
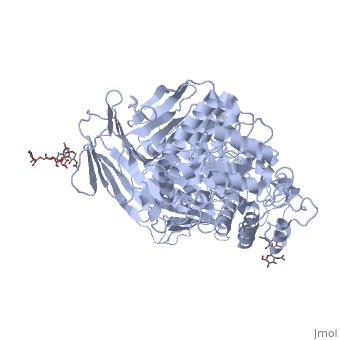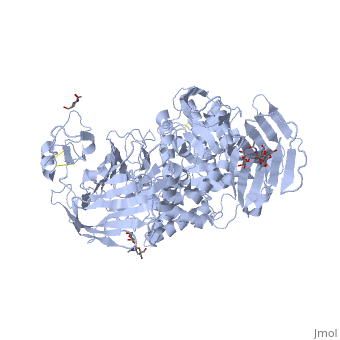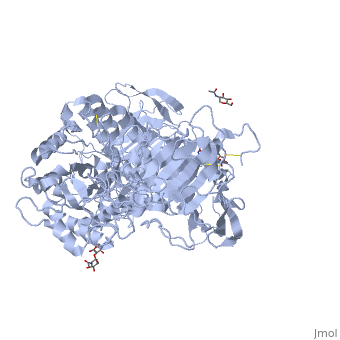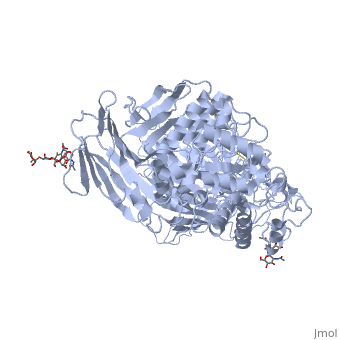
| - | [[3lpo]] - hSI N-terminal - human<br /> | ||
| - | [[3lpp]] - hSI N-terminal + kotalanol<br /> | ||
| - | [[4m56]] - BsSI + glucose - ''Bacillus subtilis''<br /> | ||
| - | [[4m8u]], [[4maz]], [[4mb1]] - BsSI (mutant)<br /> | ||
== References == | == References == | ||
<references /> | <references /> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Revision as of 08:42, 11 February 2020
| |||||||||||
References
- ↑ http://www.siumed.edu/~dking2/erg/gicells.htm
- ↑ http://www.uniprot.org/uniprot/P14410
- ↑ Maureen Barlow Pugh, ed. (2000). Stedman's Medical Dictionary (27th ed.). Baltimore, Maryland, USA: Lippincott Williams & Wilkins. p. 65. ISBN 978-0-683-40007-6.
- ↑ "SI sucrase-isomaltase (alpha-glucosidase) [Homo sapiens (human)] - Gene - NCBI"
- ↑ Naim HY, Sterchi EE, Lentze MJ (1988). "Biosynthesis of the human sucrase-isomaltase complex. Differential O-glycosylation of the sucrase subunit correlates with its position within the enzyme complex". J. Biol. Chem. 263 (15): 7242–53. PMID 3366777
- ↑ http://www.jbc.org/content/254/6/1821.full.pdf
- ↑ http://onlinelibrary.wiley.com/doi/10.1111/j.1432-1033.1977.tb11335.x/epdf
- ↑ http://www.jbc.org/content/251/11/3250.full.pdf
- ↑ http://www.iffgd.org/site/gi-disorders/other/csid
- ↑ Sim L, Willemsma C, Mohan S, Naim HY, Pinto BM, Rose DR (2010). "Structural basis for substrate selectivity in human maltase-glucoamylase and sucrase-isomaltase N-terminal domains". J. Biol. Chem. 285 (23): 17763–70. doi:10.1074/jbc.M109.078980. PMC 2878540. PMID 20356844