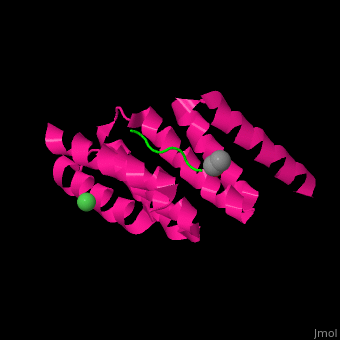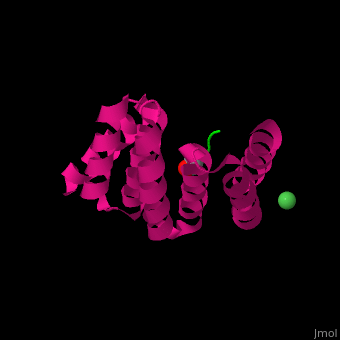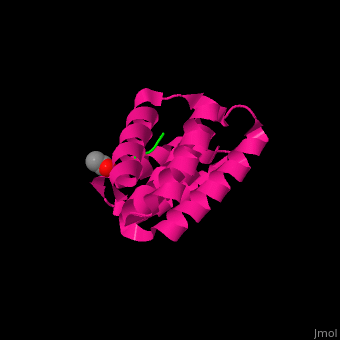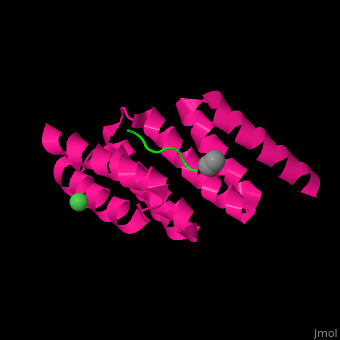This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
HOP protein
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
| - | <StructureSection load=' | + | <StructureSection load='' size='350' side='right' caption='Human HOP protein Tpr2 domain (magenta) complex with Hsp90 C-terminal pentapeptide MEEVD (green), acetyl and Ni+2 ion (green) (PDB code [[1elr]])' scene='72/725354/Cv/1'> |
== Function == | == Function == | ||
Revision as of 06:35, 20 October 2017
| |||||||||||
3D Structures of HOP protein
Updated on 20-October-2017
3fwv, 1elr – hHOP Tpr2 + Hsp90 peptide – human
1elw – hHOP Tpr1 + Hsp70 peptide
3esk – hHOP Tpr2 + Hsp70 peptide
2lni – hHOP Tpr3 - NMR
References
- ↑ Odunuga OO, Longshaw VM, Blatch GL. Hop: more than an Hsp70/Hsp90 adaptor protein. Bioessays. 2004 Oct;26(10):1058-68. PMID:15382137 doi:http://dx.doi.org/10.1002/bies.20107
- ↑ Scheufler C, Brinker A, Bourenkov G, Pegoraro S, Moroder L, Bartunik H, Hartl FU, Moarefi I. Structure of TPR domain-peptide complexes: critical elements in the assembly of the Hsp70-Hsp90 multichaperone machine. Cell. 2000 Apr 14;101(2):199-210. PMID:10786835 doi:10.1016/S0092-8674(00)80830-2