We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
Pyruvate phosphate dikinase
From Proteopedia
(Difference between revisions)
| Line 9: | Line 9: | ||
The enzyme has been found in bacteria, in C<sub>4</sub> and Crassulacean acid metabolism plants, and in parasites, but not in higher animal forms. In bacteria and parasites, PPDK functions in the direction of ATP synthesis (reminiscent of pyruvate kinase). In plants and in photosynthetic bacteria, PPDK functions in PEP formation, potentiating the rate of CO<sub>2</sub> fixation that takes place during photosynthesis. PPDK exhibits sequence homology to pyruvate phosphate synthase, and to another enzyme that utilizes phosphotransfer from PEP to a histidine residues, Enzyme I of the PEP:sugar phosphotransferase system (PTS). | The enzyme has been found in bacteria, in C<sub>4</sub> and Crassulacean acid metabolism plants, and in parasites, but not in higher animal forms. In bacteria and parasites, PPDK functions in the direction of ATP synthesis (reminiscent of pyruvate kinase). In plants and in photosynthetic bacteria, PPDK functions in PEP formation, potentiating the rate of CO<sub>2</sub> fixation that takes place during photosynthesis. PPDK exhibits sequence homology to pyruvate phosphate synthase, and to another enzyme that utilizes phosphotransfer from PEP to a histidine residues, Enzyme I of the PEP:sugar phosphotransferase system (PTS). | ||
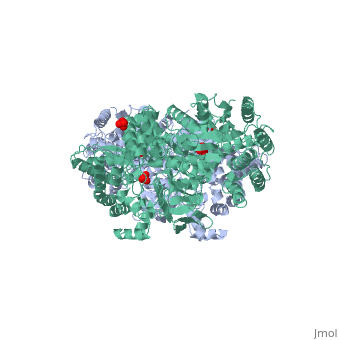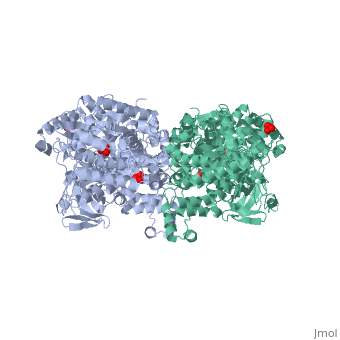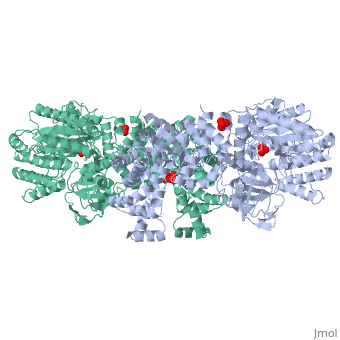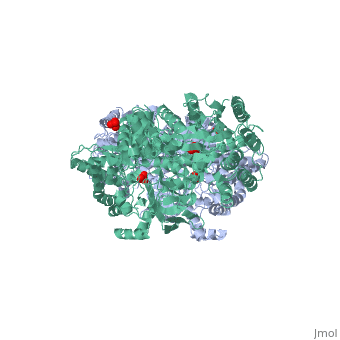
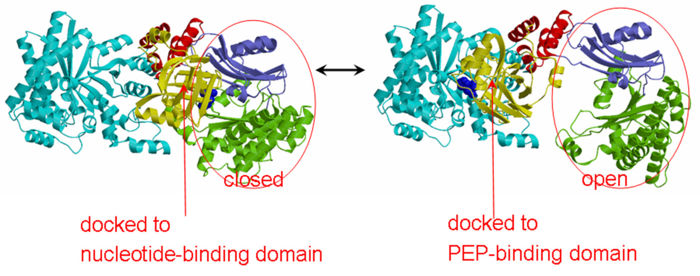
| - | PPDK assembles into homodimers of ~95 kD subunit molecular mass. The monomer is comprised of three domains and contains two distinct reaction centers located ~45 Å apart; the PEP/pyruvate partial reaction (step 1) takes place at the C-terminal domain (adopting an α/β barrel fold) and the nucleotide and inorganic phosphate partial reactions (steps 2 and 3) take place at the N-terminal domain (adopting the ATP grasp fold with two sub domains). A central domain, tethered to the N- and C-terminal domains by two closely-associated linkers, contains a phosphorylatable histidine residue (His455). To shuttle the phosphoryl group between the two reaction centers, the His-domain undergoes domain motion of ~110° swivel around the two linkers. In addition, upon detachment from the His-domain, the two nucleotide-binding sub domains undergo a ~40° hinge motion that opens the active site cleft. | + | [[Image:1KBL dimer.png|left|150px|''PPDK dimer'']]<br />PPDK assembles into homodimers of ~95 kD subunit molecular mass. The monomer is comprised of three domains and contains two distinct reaction centers located ~45 Å apart; the PEP/pyruvate partial reaction (step 1) takes place at the C-terminal domain (adopting an α/β barrel fold) and the nucleotide and inorganic phosphate partial reactions (steps 2 and 3) take place at the N-terminal domain (adopting the ATP grasp fold with two sub domains). A central domain, tethered to the N- and C-terminal domains by two closely-associated linkers, contains a phosphorylatable histidine residue (His455). To shuttle the phosphoryl group between the two reaction centers, the His-domain undergoes domain motion of ~110° swivel around the two linkers. In addition, upon detachment from the His-domain, the two nucleotide-binding sub domains undergo a ~40° hinge motion that opens the active site cleft. |
| - | + | <br>PEP-binding domain - cyan; nucleotide binding domain - green; His-domain - yellow; domain linkers -red; phosphorylatable His455 - blue spheres|200px]] | |
'''The His-domain in the two conformational states of PPDK. His455 is shown in blue spheres:'''[[Image:two_cond.jpg|right|700px]]<br><br><br> | '''The His-domain in the two conformational states of PPDK. His455 is shown in blue spheres:'''[[Image:two_cond.jpg|right|700px]]<br><br><br> | ||
Revision as of 08:10, 22 October 2017
Pyruvate Phosphate Dikinase - a Molecular Machine
| |||||||||||
3D Structures of PPDK
Updated on 22-October-2017
Proteopedia Page Contributors and Editors (what is this?)
Joel L. Sussman, Osnat Herzberg, Michal Harel, Jaime Prilusky, Alexander Berchansky, Dan Bolser, Karl Oberholser, David Canner, Eran Hodis