Anything in this section will appear adjacent to the 3D structure and will be scrollable.
Background (POSSIBLE LINKS TO OUTSIDE PAGES?)
Protein of interest: SRp20
Significance: RNA binding proteins
Organism: Homo Sapiens, Streptococcus sp. Group G
Characteristics: Ser/Arg-rich domain
Basic function: Splicing Factor
164 aa: half of it belongs to the RS (Arg/Ser) domain
MW: 19 kDA
Determined via HSQC NMR (heteronuclear single quantum coherence)
Function
General Funtions (GO INTO DETAIL ABOUT EACH)
RNA processing
Termination of transcription
Protein translation
Cellular proliferation/maturation
Involved in Insulin signaling (loss in liver results in wrong splicing of glucose metabolism genes)
Embryogenesis
Splicing Factor
The spliceosome is a complex responsible for splicing pre-mRNA to mature mRNA, composed of snRNA and associated proteins
Introns are spliced out, while exons are ligated together
Splicing is necessary for mRNA maturation, enabling it to exit the nucleus
Alternative Splicing
Alternative splicing allows many different proteins to be produced from one mRNA by splicing different combinations of introns and exons
Changes or defects in this process linked to human diseases
RNA processing factors may be targets for future therapies
SR Proteins
Characteristics:
One or two N’ term RRMs (RNA recognition motifs)
Arg/ser-rich domain downstream
Most serine residues phosphorylated
The N’ term RRM provides substrate specificity
The C’ term RS domain aids protein-protein interactions (simplifying spliceosome recruitment)
RMM
N-terminal RNA recognition motif
β1α1β2β3α2β3 Topology standard for an RRM
A four stranded β-sheet and two α-helices
Most commonly, three aromatic side-chains (in β-3 and β-1 strands) accommodate two nucleotides
Recognition enables binding of SRp20 to RNA
mRNA Maturation
Relationship to other proteins
9G8

SRp20 and 9G8 Sequence Alignment
Disease
Cancer
Oncogenic signaling pathways such as the WnT pathway cause-effect relationship with increase expression of SRp20
Downregulation of SRp20 promotes cellular senescence through AS of TP53 and generating p53 (tumor suppressor gene)
Silencing SRp20 in cancer cells: slower cell proliferation
Activator of the AS of gene CD44
Pyruvate Kinase M (PK-M gene)
Regulate Rac1b protein expression in colorectal tumor cells
Alzheimer's
TRKB: AS results in TrkB-Shc transcript
Human Papillomavirus: HPV
Increases SRp20 which in turn regulates the gene expression of HPV via interaction with A/C-rich RNA nucleobases
Overexpression of it involved in HPV-induced cancers such as cervical cancer
Structural highlights
Poor Solubility Problem
The SRp20 protein has poor solubility in its free state. This made it impossible to determine the structure of SRp20 using HSQC Spectroscopy without a modification to the free state protein. This problem was resolved by studying the proteins after fusing the RRM (RNA-recognition motif) with the immunoglobulin G-binding domain 1 of Streptococcal Protein G GB1 solubility tag.
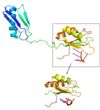
SRp20 with solubility tag
RNA Interactions
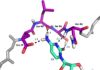
1H-15N HSQC results showed a large hydrophobic β-sheet on the RRM binds to the RNA with all four bases contacting one of the four aromatic residues” (hydrophobic interactions)
Other structural studies show that amino acids of the β-hairpin are directly hydrogen bonded to bases of nucleic acid targets
Using a smaller peptide chain reduced the NMR broadening seen with longer peptides (allowing for structure determination), though the binding affinity was also reduced
stack on Y13 in β1 and F50 in β3 (aromatic side chains), respectively.
The residue F48 (purple) inserts between the sugar rings of C1 and A2
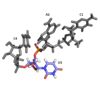
Closer look at the ligand used
Looking at the ligand
The conformation of U3 and C4 is unusual because U3 bulges out while C4 stacks over A2, partially. (IMAGE or Video showing bulge and stacking)

C4 and A2 interactions with the ligand
A2 also adopts an unusual syn conformation

A2 adopts an unusual syn conformation
C4 is maintained in its position by a hydrogen bond between C4 amino group and the A2 2’ oxygen (IMAGE)
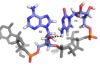
C4 is maintained in its position by a hydrogen bond between C4 amino group and the A2 2’ oxygen
RRM Domain Interactions
U3 interacts with Phe 48, Trp 40, Ala 42, and with the β2-3 loop of the RRM (IMAGE)

U3 interacts with Phe 48, Trp 40, Ala 42, and with the β2-3 loop of the RRM
C1 amino protons hydrogen bond with Leu 80 carbonyl oxygen and Glu 79 side-chain carbonyl oxygen.
C1 N3 hydrogen bonds with Asn 82 amide.
C1 O2 hydrogen bonds with Ser 81 hydroxyl group.
Ser-Arg Rich Domain
Specificity
4 nt can be accommodated by RRM β-sheet, but recognition is only
partially sequence specific.
CAUC
C1 more specific, A2 and U3 less specific
It is uncertain whether C4 is specifically recognized by the RRM
A is prefered over G at the 2 position, but no indication of preference over U or C
U3 is even less specific, could be C, G or A
The recognition of C1 is functionally necessary because a C to G mutation within the histone mRNA can impair RNA export
Advantages of low specificity
Less evolutionary pressure on bound RNA (ideal for exonic sequences)
More RNA sequences can be targeted
SRp20 can associate/disassociate with RNA more easily
Important for highly dynamic RNA metabolism processes
RNA binding affinity can be modulated by protein-protein interactions (which are dependant on the level of phosphorylation)
This can be used to tune post-transcriptional gene expression
Relevance and Conclusions
Understanding and recognizing the mechanisms that SRp20 is involved in can help find treatment and management of cancer patients
The use of SR proteins (such as SRp20) may in the future be used for targeted therapy
No structure for the C-term domain
Future Directions
Recent structural studies emphasize that not only the β-sheet surface but also the loops connecting β-strands and a-helices can be crucial for nucleic acid recognition → future research looking more closely at this possibility
Farther research could be done investigating the process of RS domain phosphorylation and how it controls splicing
Look farther into the specificity or lack of specificity relating to SR proteins
Mutate specific residues in order to map out SRp20 mechanistic function
References
Cle’ry A, Blatter M, Allain FHT. 2008. RNA recognition motifs: boring? Not quite. Current Opinion in Structural Biology 18: 290–298. Doi: 10.1016/j.sbi.2008.04.002
Corbo C, Orrù S, Salvatore F. 2013. SRp20: An overview of its role in human diseases. Biochemical and Biophysical Research Communications 436: 1–5. Doi: 10.1016/j.bbrc.2013.05.027
Hargous Y, Hautbergue GM, Tintaru AM, Skrisovska L, Golovanov AP, et. al. 2006. The EMBO Journal ) 25: 5126–5137. Doi: :10.1038/ sj.emboj.7601385