This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Kyle Burton/Sandbox1
From Proteopedia
| Line 11: | Line 11: | ||
== Structure == | == Structure == | ||
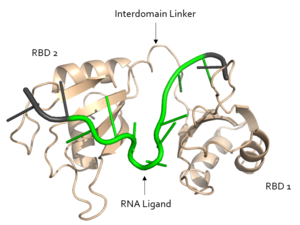
[[Image:Sex Lethal Protein Structural Overview with Labels.png|300px|right|thumb| '''Figure 2.''' Structural overview of Sxl. RNA ligand colored in green is recognized and bound, while RNA ligand colored in grey is not bound. Image created in PyMol. Structure shown is [https://www.rcsb.org/structure/1b7f PDB:1b7f].]] | [[Image:Sex Lethal Protein Structural Overview with Labels.png|300px|right|thumb| '''Figure 2.''' Structural overview of Sxl. RNA ligand colored in green is recognized and bound, while RNA ligand colored in grey is not bound. Image created in PyMol. Structure shown is [https://www.rcsb.org/structure/1b7f PDB:1b7f].]] | ||
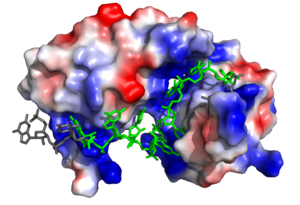
| - | Sxl is composed of two asymmetric RNA binding domains (RBD1 and RBD2) which recognize a poly-uridine site in the pre-mRNA transcript<ref name="Handa"/>. Each RBD is comprised of two alpha helices and one antiparallel four-stranded β sheet<ref name="Handa"/>. The β sheets face each other, lining the electropositive V-shaped cleft<ref name="Handa"/>. The inter-domain linker forms a distorted 3<sub>10</sub> helix which helps form the V-shaped cleft into which the pre-mRNA sequence binds<ref name="Handa"/><ref name="Black">doi: 10.1146/annurev.biochem.72.121801.161720</ref>. Sxl binds to UGUUUUUUU sequence of GUUGUUUUUUUU in tra. RBD1 binds U6-U11 and RBD2 binds U3, G4, and U5. Although the two RBDs do not interact with each other, this nine-ribonucleotide sequence must be recognized continuously to prevent U2AF from binding at the 3’ splice site<ref name="Handa"/>. The binding of Sxl to the pre-mRNA occurs in an electropositive pocket due to extensive interactions with the RNA phosphate backbone and negatively charged residues<ref name="Handa"/>. Since Sxl binds primarily with the phosphate backbone, the protein residues are not highly conserved. | + | Sxl is composed of two asymmetric RNA binding domains (RBD1 and RBD2) which recognize a poly-uridine site in the pre-mRNA transcript<ref name="Handa"/>. Each RBD is comprised of two alpha helices and one antiparallel four-stranded β sheet<ref name="Handa"/>. The β sheets face each other, lining the electropositive V-shaped cleft<ref name="Handa"/>. The inter-domain linker forms a distorted 3<sub>10</sub> helix which helps form the V-shaped cleft into which the pre-mRNA sequence binds<ref name="Handa"/><ref name="Black">doi: 10.1146/annurev.biochem.72.121801.161720</ref>. Sxl binds to UGUUUUUUU sequence of GUUGUUUUUUUU in tra. RBD1 binds U6-U11 and RBD2 binds U3, G4, and U5. Although the two RBDs do not interact with each other, this nine-ribonucleotide sequence must be recognized continuously to prevent U2AF from binding at the 3’ splice site<ref name="Handa"/>. The binding of Sxl to the pre-mRNA occurs in an electropositive pocket due to extensive interactions with the RNA phosphate backbone and negatively charged residues<ref name="Handa"/>. Since Sxl binds primarily with the phosphate backbone, the protein residues are not highly conserved. |
=== Alternative Splicing Pathways === | === Alternative Splicing Pathways === | ||
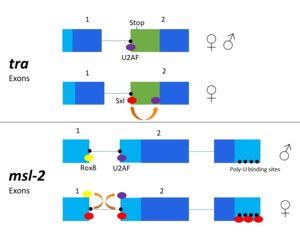
[[Image:Sxl mechanism alternativesplicing figure version2.jpg|300px|left|thumb| '''Figure 3.''' 2-dimensional representation of alternative splicing repression by Sxl on the ''tra'' and ''msl-2'' genes.]] | [[Image:Sxl mechanism alternativesplicing figure version2.jpg|300px|left|thumb| '''Figure 3.''' 2-dimensional representation of alternative splicing repression by Sxl on the ''tra'' and ''msl-2'' genes.]] | ||
| - | The alternative splicing pathways of Sxl differ, but both involve repression at the 3' splice site. The ''tra'' expression pathway only involves the 3' splice site, while the ''msl-2'' pathway involves both the 3' splice site and the 5' splice site. Both mechanisms cause U2AF binding downstream (Fig. 2)<ref name="Black"/>. | + | The alternative splicing pathways of Sxl differ, but both involve repression at the 3' splice site. The ''tra'' expression pathway only involves the 3' splice site, while the ''msl-2'' pathway involves both the 3' splice site and the 5' splice site. Both mechanisms cause U2AF binding downstream (Fig. 2)<ref name="Black"/>. U2AF is a more general splicing factor than Sxl, preferring cytidine-containing poly-uridine pre-mRNA sequences, so Sxl binds to the guanosine-containing pre-mRNA with a 10<sup>4</sup>-fold greater affinity. |
| + | |||
| + | ==== Autoregulation ==== | ||
| + | Sxl is capable of autoregulation of its own expression<ref name="Black"/>. The Sxl gene is transcribed in male flies, but the inclusion of exon 3 results in a premature stop codon, producing an inactive, truncated protein. The same Sxl promoter is active in female flies, but an additional (briefly active) Sxl promoter produces a transcript missing exon 3, thus producing an active Sxl protein<ref name="Black"/>. The active Sxl protein flanks exon 3 via multiple binding sites, surrounding the repressed exon<ref name="Black"/>. | ||
==== ''Tra'' ==== | ==== ''Tra'' ==== | ||
| Line 22: | Line 25: | ||
==== ''Msl-2'' ==== | ==== ''Msl-2'' ==== | ||
| - | === Structural | + | === Structural Basis for Recognition of Poly-U Sequences === |
The ligand pre-mRNA sequence forms a characteristic loop at U5, U6, and U7. This interaction is stabilized by π stacking between the G4 and U5 nucleotides and residues <scene name='78/783145/Aromatic_stacking/3'>Tyr 214 and Phe 256</scene>, respectively. The nucleobases are exposed to residues on Sxl due to the 2’ endo conformation of all the nucleotides except for U8, which maintains a 3’ endo conformation. | The ligand pre-mRNA sequence forms a characteristic loop at U5, U6, and U7. This interaction is stabilized by π stacking between the G4 and U5 nucleotides and residues <scene name='78/783145/Aromatic_stacking/3'>Tyr 214 and Phe 256</scene>, respectively. The nucleobases are exposed to residues on Sxl due to the 2’ endo conformation of all the nucleotides except for U8, which maintains a 3’ endo conformation. | ||
| - | + | ||
The structural interactions with regards to the targeting of the 5' splice site and of its own mRNA transcript are much less understood than the competition of Sxl with U2AF at the 3' splice site. | The structural interactions with regards to the targeting of the 5' splice site and of its own mRNA transcript are much less understood than the competition of Sxl with U2AF at the 3' splice site. | ||
| Line 65: | Line 68: | ||
== Relevance == | == Relevance == | ||
| - | As Sxl functions as a splicing repressor, it may give insight into the effects of varying mechanisms of alternate splicing both in flies and other species. Sxl may also lead to understanding of human alternative splicing factors. As an RNA binding protein, research regarding Sxl may contribute to the understanding of enzymes with RNA recognition motifs. | + | As Sxl functions as a splicing repressor, it may give insight into the effects of varying mechanisms of alternate splicing both in flies and other species. Sxl may also lead to understanding of human alternative splicing factors. As an RNA binding protein, research regarding Sxl may contribute to the understanding of enzymes with RNA recognition motifs. |
== References == | == References == | ||
<references/> | <references/> | ||
Revision as of 15:25, 29 March 2018
Contents |
Sex-Lethal Protein
| |||||||||||
Additional Reading
For more information on the U2AF splicing factor.
Relevance
As Sxl functions as a splicing repressor, it may give insight into the effects of varying mechanisms of alternate splicing both in flies and other species. Sxl may also lead to understanding of human alternative splicing factors. As an RNA binding protein, research regarding Sxl may contribute to the understanding of enzymes with RNA recognition motifs.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Handa N, Nureki O, Kurimoto K, Kim I, Sakamoto H, Shimura Y, Muto Y, Yokoyama S. Structural basis for recognition of the tra mRNA precursor by the Sex-lethal protein. Nature. 1999 Apr 15;398(6728):579-85. PMID:10217141 doi:10.1038/19242
- ↑ 2.0 2.1 Penalva LO, Sanchez L. RNA binding protein sex-lethal (Sxl) and control of Drosophila sex determination and dosage compensation. Microbiol Mol Biol Rev. 2003 Sep;67(3):343-59, table of contents. PMID:12966139
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 Black DL. Mechanisms of alternative pre-messenger RNA splicing. Annu Rev Biochem. 2003;72:291-336. doi: 10.1146/annurev.biochem.72.121801.161720., Epub 2003 Feb 27. PMID:12626338 doi:http://dx.doi.org/10.1146/annurev.biochem.72.121801.161720