This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Kyle Burton/Sandbox1
From Proteopedia
| Line 39: | Line 39: | ||
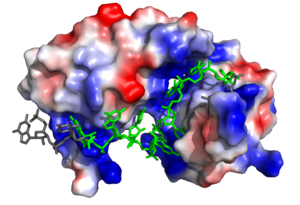
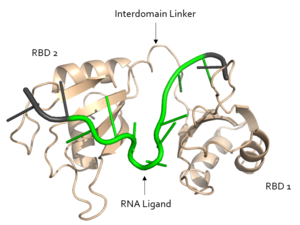
In the RNA <scene name='78/783145/U5_u6_u7_loop/2'>loop</scene>, the U7 and U8 bases are involved in π <scene name='78/783145/U7_u8_stack/1'>stacking</scene>, stabilizing the unusual 3' endo conformation of the U8 sugar<ref name="Handa"/>. U8 is further stabilized via hydrogen bonding <scene name='78/783145/U8_with_s165_and_y166/1'>interactions with Ser165 and Tyr166</scene><ref name="Handa"/>. The amine group of U8 hydrogen bonds to the the carbonyl oxygens of both Ser165 and Tyr166 <ref name="Handa"/>. | In the RNA <scene name='78/783145/U5_u6_u7_loop/2'>loop</scene>, the U7 and U8 bases are involved in π <scene name='78/783145/U7_u8_stack/1'>stacking</scene>, stabilizing the unusual 3' endo conformation of the U8 sugar<ref name="Handa"/>. U8 is further stabilized via hydrogen bonding <scene name='78/783145/U8_with_s165_and_y166/1'>interactions with Ser165 and Tyr166</scene><ref name="Handa"/>. The amine group of U8 hydrogen bonds to the the carbonyl oxygens of both Ser165 and Tyr166 <ref name="Handa"/>. | ||
| + | <scene name='78/783145/N130_interaction_with_u9/3'>U9</scene> is recognized by the interdomain linker <ref name="Handa"/>. This interaction is a salt bridge between the Asn130 side chain and a phosphate oxygen of U9. U9 is further stabilized by a second <scene name='78/783145/U9_with_interdomain_linker/1'>salt bridge</scene> between the U9 O2' and the side chain of Arg202 and the U9 O4' and the Lys197 side chain. | ||
| + | U9 facilitates the stabilization of U10, which is also recognized by the interdomain linker. <scene name='78/783145/Arg_258_interaction_w_u9_u10/3'>Arg-258 interacts with U9 and U10</scene> to form a salt bridge. | ||
| + | The O2' of U11 <scene name='78/783145/R155_intxn_with_u11/3'>interacts with Arg155</scene> to form a hydrogen bond. | ||
| - | + | The above interactions are relevant in that Sxl recognizes the specific pre-mRNA based mostly on interactions with the sugar-phosphate backbones. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
</StructureSection> | </StructureSection> | ||
Revision as of 16:44, 29 March 2018
Contents |
Sex-Lethal Protein
| |||||||||||
Additional Reading
For more information on the U2AF splicing factor.
Relevance
As Sxl functions as a splicing repressor, it may give insight into the effects of varying mechanisms of alternate splicing both in flies and other species. Sxl may also lead to understanding of human alternative splicing factors. As an RNA binding protein, research regarding Sxl may contribute to the understanding of enzymes with RNA recognition motifs.
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 Handa N, Nureki O, Kurimoto K, Kim I, Sakamoto H, Shimura Y, Muto Y, Yokoyama S. Structural basis for recognition of the tra mRNA precursor by the Sex-lethal protein. Nature. 1999 Apr 15;398(6728):579-85. PMID:10217141 doi:10.1038/19242
- ↑ 2.0 2.1 Penalva LO, Sanchez L. RNA binding protein sex-lethal (Sxl) and control of Drosophila sex determination and dosage compensation. Microbiol Mol Biol Rev. 2003 Sep;67(3):343-59, table of contents. PMID:12966139
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 Black DL. Mechanisms of alternative pre-messenger RNA splicing. Annu Rev Biochem. 2003;72:291-336. doi: 10.1146/annurev.biochem.72.121801.161720., Epub 2003 Feb 27. PMID:12626338 doi:http://dx.doi.org/10.1146/annurev.biochem.72.121801.161720
- ↑ doi: https://dx.doi.org/10.1128/mmbr.67.3.343-359.2003