This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Vpr protein
From Proteopedia
| Line 3: | Line 3: | ||
== Function == | == Function == | ||
'''Vpr protein''' (Vpr) or '''viral protein R''' is a 96 amino acid protein which encoded by the human Immunodeficiency virus type 1 ([[HIV-1]]). HIV-1 genome includes nine genes, six of them are accessory proteins, including ''vpr''. Vpr has several roles in the progression of acquired immunodeficiency syndrome ([https://en.wikipedia.org/wiki/AIDS AIDS]) disease, including regulation the HIV-1 pre-integration complex ([https://en.wikipedia.org/wiki/Pre-integration_complex PIC]) nuclear import and virus replication in non-dividing macrophages<ref name='Morellet2003'>PMID:12614620</ref>. Moreover, Vpr prevents mitosis of dividing infect cells by blockade at G2 phase, when the viral promoter is more active, and induced apoptosis of these cells in G1 and M phase. Both the activities of cell cycle arrest and induce apoptosis has proven to be independence by mutations that can cause only one of these affects<ref name='Gonzalez2017'>PMID:28075409</ref>. | '''Vpr protein''' (Vpr) or '''viral protein R''' is a 96 amino acid protein which encoded by the human Immunodeficiency virus type 1 ([[HIV-1]]). HIV-1 genome includes nine genes, six of them are accessory proteins, including ''vpr''. Vpr has several roles in the progression of acquired immunodeficiency syndrome ([https://en.wikipedia.org/wiki/AIDS AIDS]) disease, including regulation the HIV-1 pre-integration complex ([https://en.wikipedia.org/wiki/Pre-integration_complex PIC]) nuclear import and virus replication in non-dividing macrophages<ref name='Morellet2003'>PMID:12614620</ref>. Moreover, Vpr prevents mitosis of dividing infect cells by blockade at G2 phase, when the viral promoter is more active, and induced apoptosis of these cells in G1 and M phase. Both the activities of cell cycle arrest and induce apoptosis has proven to be independence by mutations that can cause only one of these affects<ref name='Gonzalez2017'>PMID:28075409</ref>. | ||
| + | |||
| + | === Vpr induce cell cycle arrest by recruits cellular targets for degradation<ref>PMID:27571178</ref> === | ||
| + | One phenotype of Vpr expression is the induction of cell cycle arrest in G2 phase. This arrest executes by engaging of DNA damage–binding protein 1 (DDB1) and CUL4A-associated factor 1 (DCAF1) to Vpr. DCAF1 is a substrate receptor of the Cullin4–RING E3 ubiquitin ligase (CRL4) of the host ubiquitin–proteasome-mediated protein degradation pathway. Mutations in Vpr that abolish its interaction with DCAF1, or silencing of DCAF1, eliminate cell-cycle arrest. | ||
== Disease == | == Disease == | ||
Revision as of 00:11, 8 April 2018
| |||||||||||
3D Structures of Vpr protein
Updated on 08-April-2018
1m8l – Vpr - NMR - HIV-1
1esx - synthetic Vpr - NMR - HIV-1
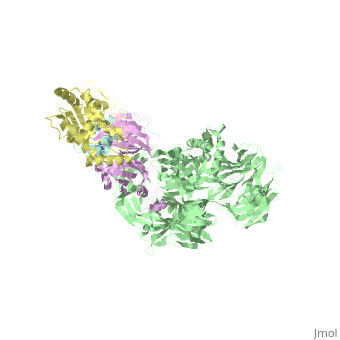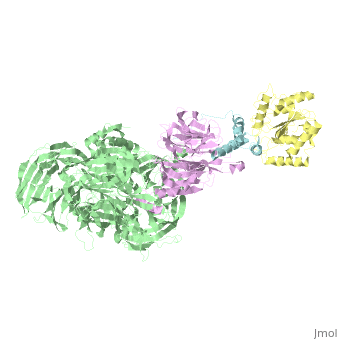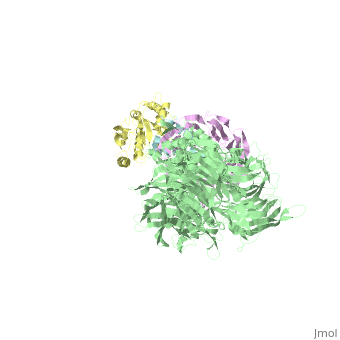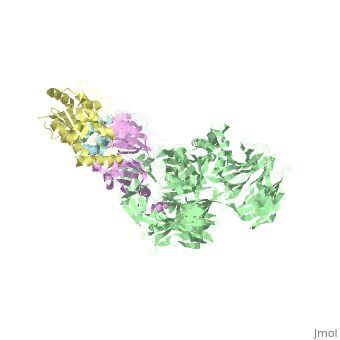
5jk7 – Vpr + DDB1 + DCAF-1 + UNG2 – X-ray solution - HIV-1
1x9v – Dimeric structure of the Vpr C-terminal domain - NMR
1vpc - C-terminal domain of Vpr - NMR - HIV-1
1fi0 - Vpr residues 13-33 in micelles - NMR - HIV-1
1bde - NMR solution of Vpr peptides connected to cell cycle arrest and nuclear provirus transfer
5b56 - Importin subunit alpha-1 + Vpr C-terminal domain - crystallographic analysis
1kzs, 1kzt, 1kzv - Vpr residues 34-51 - NMR - HIV-1
1dsj - Vpr residues 50-75 - NMR - HIV-1
1ceu - Vpr N-terminal domain - NMR - HIV-1
1dsk - Vpr residues 59-86 - NMR - HIV-1
References
- ↑ 1.0 1.1 Morellet N, Bouaziz S, Petitjean P, Roques BP. NMR structure of the HIV-1 regulatory protein VPR. J Mol Biol. 2003 Mar 14;327(1):215-27. PMID:12614620
- ↑ 2.0 2.1 Gonzalez ME. The HIV-1 Vpr Protein: A Multifaceted Target for Therapeutic Intervention. Int J Mol Sci. 2017 Jan 10;18(1). pii: ijms18010126. doi: 10.3390/ijms18010126. PMID:28075409 doi:http://dx.doi.org/10.3390/ijms18010126
- ↑ Wu Y, Zhou X, Barnes CO, DeLucia M, Cohen AE, Gronenborn AM, Ahn J, Calero G. The DDB1-DCAF1-Vpr-UNG2 crystal structure reveals how HIV-1 Vpr steers human UNG2 toward destruction. Nat Struct Mol Biol. 2016 Aug 29. doi: 10.1038/nsmb.3284. PMID:27571178 doi:http://dx.doi.org/10.1038/nsmb.3284
- ↑ 4.0 4.1 Emerman M. HIV-1, Vpr and the cell cycle. Curr Biol. 1996 Sep 1;6(9):1096-103. PMID:8805364