This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Alexis Neyman/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 9: | Line 9: | ||
== Structure == | == Structure == | ||
| - | [[Image:2D_RRM_and_RS_SRp20_with_fun_shapes3.png|250 px|right|thumb|Figure | + | [[Image:2D_RRM_and_RS_SRp20_with_fun_shapes3.png|250 px|right|thumb|Figure 1: SRp20 RRM and RS domains are shown.]] The structure of SRp20 was determined by heteronuclear single quantum coherence ([https://en.wikipedia.org/wiki/Heteronuclear_single_quantum_coherence_spectroscopy HSQC]) NMR. The structure is composed of one RNA recognition motif (RRM) at the N-terminus and one Arg/Ser (AR) domain at the C-terminus where the Ser residues are phosphorylated<ref name="corbo">PMID:23685143</ref>. The RRM of SRp20 demonstrates the β1α1β2β3α2β3 topology seen in other [https://en.wikipedia.org/wiki/RNA_recognition_motif RRMs]. The role of the RRM region is to provide substrate specificity where SRp20 interacts with splicing enhancing sequences in mRNA. There have been no determined 3D structures of the RS domain thus it is unclear what its exact role is. However there have been some speculation that it might be involved in aiding protein-protein interactions in the spliceosome. It contains 164 amino acids, half belonging to the RRM and other half to the RS domain (Figure 1). SRp20 has a molecular weight of 19 kDA<ref name="corbo">PMID:23685143</ref>. |
=== Poor Solubility Problem === | === Poor Solubility Problem === | ||
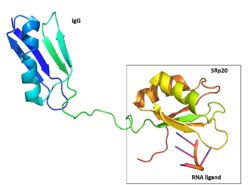
| - | The SRp20 protein has poor [https://en.wikipedia.org/wiki/Solubility solubility] in its free state. [[Image:Figure_Three_Solubility_Tag_Edited.jpg|250 px|right|thumb|Figure | + | The SRp20 protein has poor [https://en.wikipedia.org/wiki/Solubility solubility] in its free state. [[Image:Figure_Three_Solubility_Tag_Edited.jpg|250 px|right|thumb|Figure 2: SRp20 with the solubility IgG tag and the RNA ligand are shown. The solubility tag is at the N' terminus, in front of the RRM. While in the actual protein, the RRM is at the N'terminus, in front of the RS domain. Image created using ''Pymol'']]This made it impossible to determine the structure of SRp20 using HSQC Spectroscopy without a modification to the free state protein. This problem was resolved by studying the proteins after fusing the RRM (RNA-recognition motif) with the immunoglobulin G-binding domain 1 of Streptococcal [https://en.wikipedia.org/wiki/Protein_G Protein G] GB1 solubility tag (Figure 2) <ref name="Hargous">PMID:17036044</ref>. |
=== RNA Interactions === | === RNA Interactions === | ||
| - | 1H-15N HSQC results showed a large hydrophobic β-sheet on the RRM binding to the RNA with all four bases interacting with one of the four aromatic residues via hydrophobic interactions <ref name="Hargous">PMID:17036044</ref>. [https://en.wikipedia.org/wiki/Beta_hairpin β-hairpin] amino acids are hydrogen bonded to bases on nucleic acid targets <ref name="Clery">PMID:18515081</ref>. This suggests that the β-hairpin plays a role in SRp20 selectivity for specific ligands. The researchers used a smaller peptide chain to reduce the NMR broadening seen with longer peptides (allowing for structure determination), with the consequence of reduced binding affinity. The ligand used was <scene name='78/781963/Looking_at_the_ligand/1'>CAUC</scene>. The conformation of U3 and C4 shows that U3 bulges out while C4 partially stacks over A2. Interactions with the RRM that the researchers saw were that <scene name='78/781963/C1_and_tyr_13/1'>C1 stacks with Y13</scene> in β1 and <scene name='78/781963/A2_phe_50/1'>A2 stacks with F50</scene> in β3. These aromatic side chains form hydrophobic interactions with the ligand when stacked (Figure | + | 1H-15N HSQC results showed a large hydrophobic β-sheet on the RRM binding to the RNA with all four bases interacting with one of the four aromatic residues via hydrophobic interactions <ref name="Hargous">PMID:17036044</ref>. [https://en.wikipedia.org/wiki/Beta_hairpin β-hairpin] amino acids are hydrogen bonded to bases on nucleic acid targets <ref name="Clery">PMID:18515081</ref>. This suggests that the β-hairpin plays a role in SRp20 selectivity for specific ligands. The researchers used a smaller peptide chain to reduce the NMR broadening seen with longer peptides (allowing for structure determination), with the consequence of reduced binding affinity. The ligand used was <scene name='78/781963/Looking_at_the_ligand/1'>CAUC</scene>. The conformation of U3 and C4 shows that U3 bulges out while C4 partially stacks over A2. Interactions with the RRM that the researchers saw were that <scene name='78/781963/C1_and_tyr_13/1'>C1 stacks with Y13</scene> in β1 and <scene name='78/781963/A2_phe_50/1'>A2 stacks with F50</scene> in β3. These aromatic side chains form hydrophobic interactions with the ligand when stacked (Figure 3). Also, the residue <scene name='78/781963/C1_a2_phe48/1'>F48 inserts between the sugar rings of C1 and A2</scene>. <<scene name='78/781963/C1_binding_pocket3/1'>scene name='78/781963/C1_binding_pocket/1</scene>C1 is recognized definitively by the RRM</scene>. The C1 amino proton hydrogen bonds with the Leu 80 carbonyl oxygen and the Glu 79 side-chain carbonyl oxygen in SRp20. The C1 N3 hydrogen bonds with the Asn 82 amide. The C1 O2 hydrogen bonds with the Ser 81 hydroxyl group <ref name="Hargous">PMID:17036044</ref>. |
| - | It was also noted that <scene name='78/781963/A2_syn_conformation/1'>A2</scene> adopts an unusual syn conformation. U3 interacts with <scene name='78/781963/U3_hydrophobic_interactions/2'>Phe 48, Trp 40, Ala 42,</scene> and with the β2-3 loop of the RRM. These residues are all hydrophobic, offering a large hydrophobic surface that helps bind the ligand, as well as prevents the solvent from binding. Additionally, C4 is maintained in its position by a <scene name='78/781963/C4_a2_h_bond/1'>hydrogen bond between C4 amino group and the A2 2’ oxygen</scene> <ref name="Hargous">PMID:17036044</ref>. [[Image:Figure_4_C1_and_A2_interactions_Edited2.png|300 px|left|thumb|Figure | + | It was also noted that <scene name='78/781963/A2_syn_conformation/1'>A2</scene> adopts an unusual syn conformation. U3 interacts with <scene name='78/781963/U3_hydrophobic_interactions/2'>Phe 48, Trp 40, Ala 42,</scene> and with the β2-3 loop of the RRM. These residues are all hydrophobic, offering a large hydrophobic surface that helps bind the ligand, as well as prevents the solvent from binding. Additionally, C4 is maintained in its position by a <scene name='78/781963/C4_a2_h_bond/1'>hydrogen bond between C4 amino group and the A2 2’ oxygen</scene> <ref name="Hargous">PMID:17036044</ref>. [[Image:Figure_4_C1_and_A2_interactions_Edited2.png|300 px|left|thumb|Figure 3: C1 and A2 on the RNA ligand interacting with hydrophobic residues (Tyr 13, Phe 50, Phe 48) in the RRM domain of the SRp20 protein. Image created using ''Pymol''.]] |
=== RRM Stability=== | === RRM Stability=== | ||
| - | SRp20 has a <scene name='78/781963/Hydrophobic_core/1'>hydrophobic core</scene>, which may contribute to the stability of the protein. A previous study, looking at the RRM in [https://en.wikipedia.org/wiki/TARDBP TDP-43] has suggested that the hydrophobic core may be a strong contributing factor to the protein’s stability | + | SRp20 has a <scene name='78/781963/Hydrophobic_core/1'>hydrophobic core</scene>, which may contribute to the stability of the protein. A previous study, looking at the RRM in [https://en.wikipedia.org/wiki/TARDBP TDP-43] has suggested that the hydrophobic core may be a strong contributing factor to the protein’s stability <ref>PMID:24497641</ref>. In a different study, it was determined that, in the U11/U12-65K protein, the β-sheet packs against the two α-helices by way of hydrophobic interactions and that the resulting stabilization could be critical for the proper folding and orientation of elements for RNA binding <ref>PMID:19447915</ref>. Due to the conservative nature of RRMs, it could be speculated that the hydrophobic core found in SRp20, between the β-sheet and two α-helices, could contribute to the stability of its RRM in a similar fashion. However, additional studies done specifically on SRp20 need to be done to confirm this supposition. |
===RRM Specificity=== | ===RRM Specificity=== | ||
| Line 32: | Line 32: | ||
== Relationship to 9G8 == | == Relationship to 9G8 == | ||
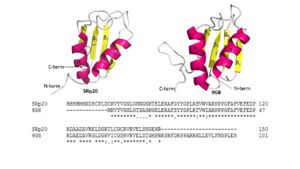
| - | [[Image:Combined_SRp20_and_9G8_Image.jpg|300 px|left|thumb|Figure | + | [[Image:Combined_SRp20_and_9G8_Image.jpg|300 px|left|thumb|Figure 4: Comparing SRp20 and 9G8 RRMs and sequence alignments. Structural images created using ''Pymol'']] <scene name='78/781963/Rrm_motif/1'>SRp20</scene> and splicing factor <scene name='78/781963/9g8_rrm/1'>9G8</scene> are both sequence specific RNA binding proteins (Figure 4). They are the smallest members of the Serine-and-Arginine Rich (SR) protein family. Both RNA Recognition Motifs (RRMs) have a similar βαββαβ topology. SRp20 and 9G8 are 80% identical. The sequence alignment shows the alignment of the RRMs of SRp20 and 9G8 <ref name="Hargous">PMID:17036044</ref>. SRp20 binds pyrimidine rich areas while 9G8 binds purine rich areas.This difference in binding comes from the fact that 9G8 has a [https://en.wikipedia.org/wiki/Zinc_finger zinc knuckle] that recognizes GAC triplets <ref name="Cava">PMID:10094314 </ref>. 9G8s RRM is followed by a zinc knuckle and then the SR domain whereas SRp20s RRM is followed directly by the SR domain. When 9G8 lacks a zinc knuckle, it binds pyrimidine-rich sequences like SRp20 <ref name="Hargous">PMID:17036044</ref>. The zinc knuckle of 9G8 contains glycine residues at positions 5 and 8 and charged residues at positions 6 and 13 that are highly conserved <ref name="Cava">PMID:10094314 </ref>. Due to the poor solubility problem, a structure for the zinc knuckle of 9G8 is not available to show in an image. |
A study by Huang and Steitz showed that 9G8 and SRp20 promote the export of mRNA from the nucleus<ref name="HS">PMID:11336712 </ref>. There is a 101-nt sequence in the coding region of mouse histone H2a mRNA that promotes the the export of intronless human β-globin [https://en.wikipedia.org/wiki/Complementary_DNA cDNA] transcripts <ref name="HC">PMID:9294170 </ref>. Of this 101-nt sequence, there is specifically a 22-nt sequence that is necessary for export activity. UV [https://en.wikipedia.org/wiki/Cross-link cross-linking] and [https://en.wikipedia.org/wiki/Immunoprecipitation immunoprecipitation] experiments determined SRp20 and 9G8 specifically associating with the 22-nt sequence. SR proteins were associating with the 22-nt sequence by adding antibodies specific for SRp20 then 9G8. SRp20 antibodies inhibited mRNA export 3-fold while 9G8 antibodies inhibited mRNA export at least 10-fold showing that SRp20 and 9G8 are active factors that promote mRNA export. It was shown that SRp20 and 9G8 are cross-linked to polyadenylated RNA in the nucleus and cytoplasm showing that both proteins play a direct role in mRNA export from the nucleus <ref name="HS">PMID:11336712 </ref>. | A study by Huang and Steitz showed that 9G8 and SRp20 promote the export of mRNA from the nucleus<ref name="HS">PMID:11336712 </ref>. There is a 101-nt sequence in the coding region of mouse histone H2a mRNA that promotes the the export of intronless human β-globin [https://en.wikipedia.org/wiki/Complementary_DNA cDNA] transcripts <ref name="HC">PMID:9294170 </ref>. Of this 101-nt sequence, there is specifically a 22-nt sequence that is necessary for export activity. UV [https://en.wikipedia.org/wiki/Cross-link cross-linking] and [https://en.wikipedia.org/wiki/Immunoprecipitation immunoprecipitation] experiments determined SRp20 and 9G8 specifically associating with the 22-nt sequence. SR proteins were associating with the 22-nt sequence by adding antibodies specific for SRp20 then 9G8. SRp20 antibodies inhibited mRNA export 3-fold while 9G8 antibodies inhibited mRNA export at least 10-fold showing that SRp20 and 9G8 are active factors that promote mRNA export. It was shown that SRp20 and 9G8 are cross-linked to polyadenylated RNA in the nucleus and cytoplasm showing that both proteins play a direct role in mRNA export from the nucleus <ref name="HS">PMID:11336712 </ref>. | ||
Revision as of 15:40, 10 April 2018
Biological Structure of SRp20
| |||||||||||