This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
AppA protein BLUF domain
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
==Domains== | ==Domains== | ||
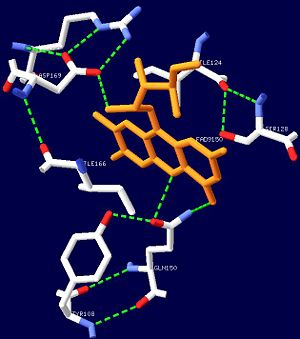
[[Image:FADbonding.jpg|thumb|left|300px|Figure 1. The hydrogen bonds the FAD ligand forms with nearby amino acid residues.]] | [[Image:FADbonding.jpg|thumb|left|300px|Figure 1. The hydrogen bonds the FAD ligand forms with nearby amino acid residues.]] | ||
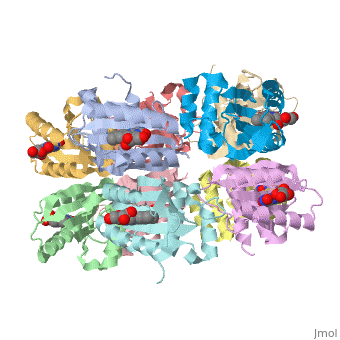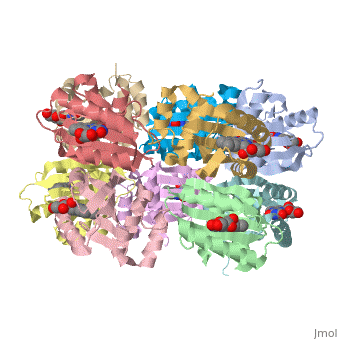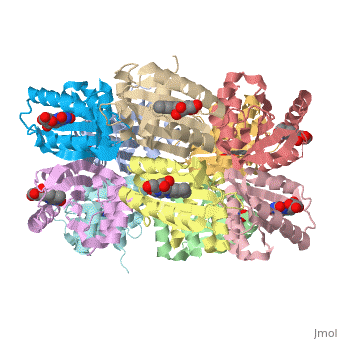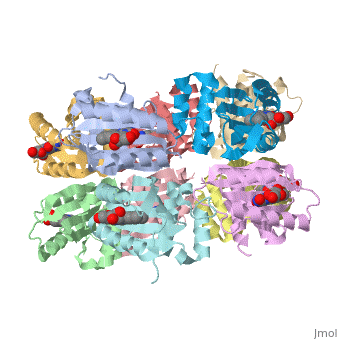
| - | The BLUF domain is a <scene name=' | + | The BLUF domain is a <scene name='44/447813/Cv/15'>decamer</scene> with a molecular weight of approximately 160kDa<ref name="one" />. There are ten monomers observed in each asymmetric unit. The crystalline structure of the BLUF domain from the T110078 protein was solved by single isomorphous replacement (SIR) method using a mercury derivative. The <scene name='44/447813/Cv/17'>double ringed decamer has a diameter of approximately 95Å</scene>, a <scene name='44/447813/Cv/18'>thickness of 60Å</scene> and a <scene name='44/447813/Cv/19'>central channel approximately 35Å in diameter</scene><ref name="one" />. |
Each monomer is comprised of 5 <scene name='44/447813/Cv/12'>β-strands</scene> and 4 <scene name='44/447813/Cv/13'>α-helices</scene> in the order of β1α1β2β3α2β4β5α3α4. Specifically, the BLUF domain of the monomer contains β1α1β2β3α2β4β5, while the C-terminal domain contains α3α4<ref name="one" />. The C-terminal domain interacts with the end of the β-sheet of the neighbouring monomer. | Each monomer is comprised of 5 <scene name='44/447813/Cv/12'>β-strands</scene> and 4 <scene name='44/447813/Cv/13'>α-helices</scene> in the order of β1α1β2β3α2β4β5α3α4. Specifically, the BLUF domain of the monomer contains β1α1β2β3α2β4β5, while the C-terminal domain contains α3α4<ref name="one" />. The C-terminal domain interacts with the end of the β-sheet of the neighbouring monomer. | ||
Revision as of 13:48, 24 May 2018
| |||||||||||
3D structures of BLUF domain protein
Updated on 24-May-2018
2byc - RsBLUF dark structure - Rhodobacter sphaeroides
2iyg, 2iyi - RsBLUF dark structure BLUF domain (mutant)
1x0p - BLUF - Thermosynechococcus elongatus
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 Kita A, Okajima K, Morimoto Y, Ikeuchi M, Miki K. Structure of a cyanobacterial BLUF protein, Tll0078, containing a novel FAD-binding blue light sensor domain. J Mol Biol. 2005 May 27;349(1):1-9. Epub 2005 Apr 9. PMID:15876364 doi:10.1016/j.jmb.2005.03.067
- ↑ van der Horst MA, Hellingwerf KJ. Photoreceptor proteins, "star actors of modern times": a review of the functional dynamics in the structure of representative members of six different photoreceptor families. Acc Chem Res. 2004 Jan;37(1):13-20. PMID:14730990 doi:10.1021/ar020219d
- ↑ 3.0 3.1 Masuda S, Bauer CE. AppA is a blue light photoreceptor that antirepresses photosynthesis gene expression in Rhodobacter sphaeroides. Cell. 2002 Sep 6;110(5):613-23. PMID:12230978
- ↑ Laan W, van der Horst MA, van Stokkum IH, Hellingwerf KJ. Initial characterization of the primary photochemistry of AppA, a blue-light-using flavin adenine dinucleotide-domain containing transcriptional antirepressor protein from Rhodobacter sphaeroides: a key role for reversible intramolecular proton transfer from the flavin adenine dinucleotide chromophore to a conserved tyrosine? Photochem Photobiol. 2003 Sep;78(3):290-7. PMID:14556317
- ↑ Hasegawa K, Masuda S, Ono TA. Spectroscopic analysis of the dark relaxation process of a photocycle in a sensor of blue light using FAD (BLUF) protein Slr1694 of the cyanobacterium Synechocystis sp. PCC6803. Plant Cell Physiol. 2005 Jan;46(1):136-46. Epub 2005 Jan 19. PMID:15659451 doi:10.1093/pcp/pci003
- ↑ 6.0 6.1 Iseki M, Matsunaga S, Murakami A, Ohno K, Shiga K, Yoshida K, Sugai M, Takahashi T, Hori T, Watanabe M. A blue-light-activated adenylyl cyclase mediates photoavoidance in Euglena gracilis. Nature. 2002 Feb 28;415(6875):1047-51. PMID:11875575 doi:10.1038/4151047a
- ↑ Yuan H, Anderson S, Masuda S, Dragnea V, Moffat K, Bauer C. Crystal structures of the Synechocystis photoreceptor Slr1694 reveal distinct structural states related to signaling. Biochemistry. 2006 Oct 24;45(42):12687-94. PMID:17042486 doi:10.1021/bi061435n
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Amanda Cookhouse, Jaime Prilusky