This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
2c6n
From Proteopedia
| Line 1: | Line 1: | ||
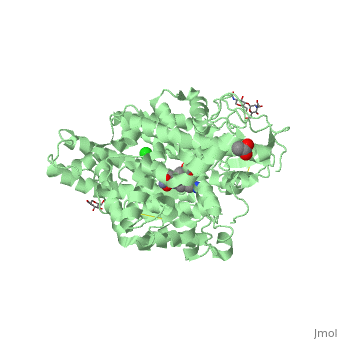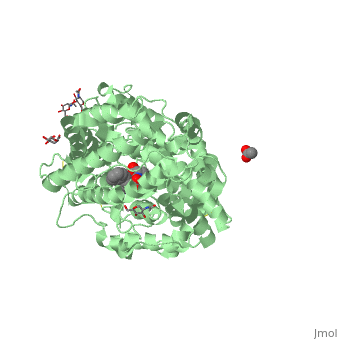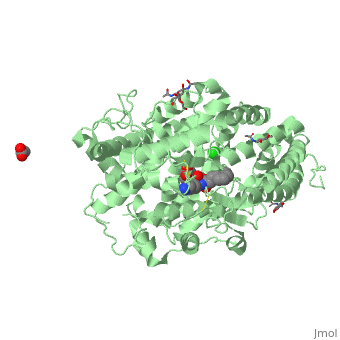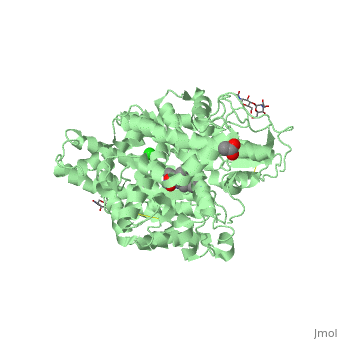
==Structure of human somatic angiontensin-I converting enzyme N domain with lisinopril== | ==Structure of human somatic angiontensin-I converting enzyme N domain with lisinopril== | ||
| - | <StructureSection load='2c6n' size='340' side='right' caption='[[2c6n]], [[Resolution|resolution]] 3.00Å' scene=''> | + | <StructureSection load='2c6n' size='340' side='right'caption='[[2c6n]], [[Resolution|resolution]] 3.00Å' scene=''> |
== Structural highlights == | == Structural highlights == | ||
| - | <table><tr><td colspan='2'>[[2c6n]] is a 2 chain structure with sequence from [http://en.wikipedia.org/wiki/Human Human]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2C6N OCA]. For a <b>guided tour on the structure components</b> use [http:// | + | <table><tr><td colspan='2'>[[2c6n]] is a 2 chain structure with sequence from [http://en.wikipedia.org/wiki/Human Human]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2C6N OCA]. For a <b>guided tour on the structure components</b> use [http://proteopedia.org/fgij/fg.htm?mol=2C6N FirstGlance]. <br> |
| - | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=ACT:ACETATE+ION'>ACT</scene>, <scene name='pdbligand=CL:CHLORIDE+ION'>CL</scene>, <scene name='pdbligand=GOL:GLYCEROL'>GOL</scene>, <scene name='pdbligand=LPR:[N2-[(S)-1-CARBOXY-3-PHENYLPROPYL]-L-LYSYL-L-PROLINE'>LPR</scene>, <scene name='pdbligand=NAG:N-ACETYL-D-GLUCOSAMINE'>NAG</scene>, <scene name='pdbligand=ZN:ZINC+ION'>ZN</scene></td></tr> | + | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=ACT:ACETATE+ION'>ACT</scene>, <scene name='pdbligand=CL:CHLORIDE+ION'>CL</scene>, <scene name='pdbligand=GOL:GLYCEROL'>GOL</scene>, <scene name='pdbligand=LPR:[N2-[(S)-1-CARBOXY-3-PHENYLPROPYL]-L-LYSYL-L-PROLINE'>LPR</scene>, <scene name='pdbligand=NAG:N-ACETYL-D-GLUCOSAMINE'>NAG</scene>, <scene name='pdbligand=ZN:ZINC+ION'>ZN</scene></td></tr> |
<tr id='related'><td class="sblockLbl"><b>[[Related_structure|Related:]]</b></td><td class="sblockDat">[[2c6f|2c6f]]</td></tr> | <tr id='related'><td class="sblockLbl"><b>[[Related_structure|Related:]]</b></td><td class="sblockDat">[[2c6f|2c6f]]</td></tr> | ||
<tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[http://en.wikipedia.org/wiki/Peptidyl-dipeptidase_A Peptidyl-dipeptidase A], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=3.4.15.1 3.4.15.1] </span></td></tr> | <tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[http://en.wikipedia.org/wiki/Peptidyl-dipeptidase_A Peptidyl-dipeptidase A], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=3.4.15.1 3.4.15.1] </span></td></tr> | ||
| - | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[http:// | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[http://proteopedia.org/fgij/fg.htm?mol=2c6n FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2c6n OCA], [http://pdbe.org/2c6n PDBe], [http://www.rcsb.org/pdb/explore.do?structureId=2c6n RCSB], [http://www.ebi.ac.uk/pdbsum/2c6n PDBsum], [http://prosat.h-its.org/prosat/prosatexe?pdbcode=2c6n ProSAT]</span></td></tr> |
</table> | </table> | ||
== Disease == | == Disease == | ||
| Line 35: | Line 35: | ||
==See Also== | ==See Also== | ||
*[[ACE Inhibitor Lisinopril|ACE Inhibitor Lisinopril]] | *[[ACE Inhibitor Lisinopril|ACE Inhibitor Lisinopril]] | ||
| - | *[[Angiotensin-Converting Enzyme|Angiotensin-Converting Enzyme]] | + | *[[Angiotensin-Converting Enzyme 3D structures|Angiotensin-Converting Enzyme 3D structures]] |
== References == | == References == | ||
<references/> | <references/> | ||
| Line 41: | Line 41: | ||
</StructureSection> | </StructureSection> | ||
[[Category: Human]] | [[Category: Human]] | ||
| + | [[Category: Large Structures]] | ||
[[Category: Peptidyl-dipeptidase A]] | [[Category: Peptidyl-dipeptidase A]] | ||
[[Category: Acharya, K R]] | [[Category: Acharya, K R]] | ||
Revision as of 07:28, 29 April 2020
Structure of human somatic angiontensin-I converting enzyme N domain with lisinopril
| |||||||||||
Categories: Human | Large Structures | Peptidyl-dipeptidase A | Acharya, K R | Corradi, H R | Nichinda, A | Schwager, S L.U | Sturrock, E D | Angiotensin | Angiotensin-i converting enzyme | Carboxypeptidase | Glycoprotein | Hydrolase | Hydrolase-hydrolase inhibitor complex | Lisinopril | Metal-binding | Metalloprotease | N domain | Phosphorylation | Protease | Transmembrane | Zinc metallopeptidase