|
|
| Line 1: |
Line 1: |
| | | | |
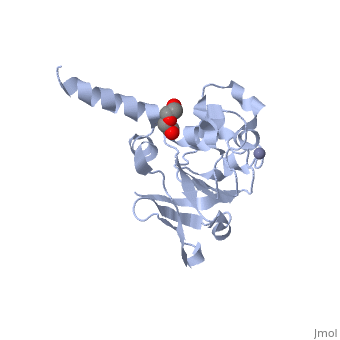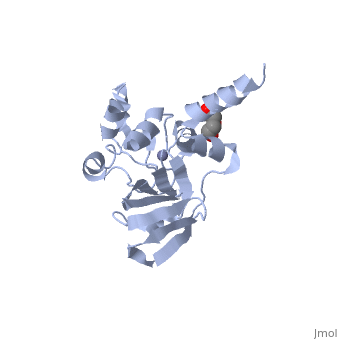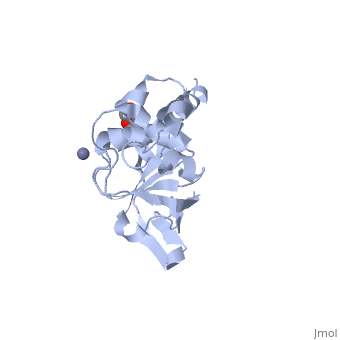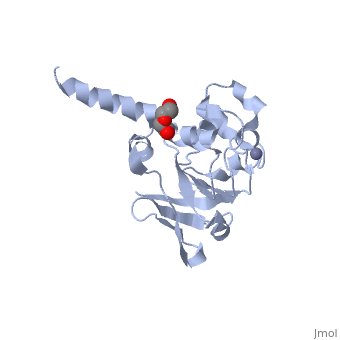
| | ==Crystal structure of 3-isopropylmalate dehydratase (leuD)from Methhanocaldococcus Jannaschii DSM2661 (MJ1271)== | | ==Crystal structure of 3-isopropylmalate dehydratase (leuD)from Methhanocaldococcus Jannaschii DSM2661 (MJ1271)== |
| - | <StructureSection load='2pkp' size='340' side='right' caption='[[2pkp]], [[Resolution|resolution]] 2.10Å' scene=''> | + | <StructureSection load='2pkp' size='340' side='right'caption='[[2pkp]], [[Resolution|resolution]] 2.10Å' scene=''> |
| | == Structural highlights == | | == Structural highlights == |
| - | <table><tr><td colspan='2'>[[2pkp]] is a 1 chain structure with sequence from [http://en.wikipedia.org/wiki/Metja Metja]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2PKP OCA]. For a <b>guided tour on the structure components</b> use [http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=2PKP FirstGlance]. <br> | + | <table><tr><td colspan='2'>[[2pkp]] is a 1 chain structure with sequence from [https://en.wikipedia.org/wiki/Metja Metja]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2PKP OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=2PKP FirstGlance]. <br> |
| - | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=PEG:DI(HYDROXYETHYL)ETHER'>PEG</scene>, <scene name='pdbligand=ZN:ZINC+ION'>ZN</scene></td></tr> | + | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=PEG:DI(HYDROXYETHYL)ETHER'>PEG</scene>, <scene name='pdbligand=ZN:ZINC+ION'>ZN</scene></td></tr> |
| - | <tr id='gene'><td class="sblockLbl"><b>[[Gene|Gene:]]</b></td><td class="sblockDat">aksE ([http://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Info&srchmode=5&id=243232 METJA])</td></tr> | + | <tr id='gene'><td class="sblockLbl"><b>[[Gene|Gene:]]</b></td><td class="sblockDat">aksE ([https://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Info&srchmode=5&id=243232 METJA])</td></tr> |
| - | <tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[http://en.wikipedia.org/wiki/Methanogen_homoaconitase Methanogen homoaconitase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=4.2.1.114 4.2.1.114] </span></td></tr> | + | <tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[https://en.wikipedia.org/wiki/Methanogen_homoaconitase Methanogen homoaconitase], with EC number [https://www.brenda-enzymes.info/php/result_flat.php4?ecno=4.2.1.114 4.2.1.114] </span></td></tr> |
| - | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=2pkp FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2pkp OCA], [http://pdbe.org/2pkp PDBe], [http://www.rcsb.org/pdb/explore.do?structureId=2pkp RCSB], [http://www.ebi.ac.uk/pdbsum/2pkp PDBsum], [http://prosat.h-its.org/prosat/prosatexe?pdbcode=2pkp ProSAT], [http://www.topsan.org/Proteins/RSGI/2pkp TOPSAN]</span></td></tr> | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=2pkp FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2pkp OCA], [https://pdbe.org/2pkp PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=2pkp RCSB], [https://www.ebi.ac.uk/pdbsum/2pkp PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=2pkp ProSAT], [https://www.topsan.org/Proteins/RSGI/2pkp TOPSAN]</span></td></tr> |
| | </table> | | </table> |
| | == Function == | | == Function == |
| - | [[http://www.uniprot.org/uniprot/HACB_METJA HACB_METJA]] Hydro-lyase with broad substrate specificity for cis-unsaturated tricarboxylic acids. Catalyzes both the reversible dehydration of (R)-homocitrate ((R)-2-hydroxybutane-1,2,4-tricarboxylate) to produce cis-homoaconitate ((Z)-but-1-ene-1,2,4-tricarboxylate), and its hydration to homoisocitrate ((1R,2S)-1-hydroxybutane-1,2,4-tricarboxylate). Is also able to hydrate the analogous longer chain substrates cis-homo(2)-aconitate, cis-homo(3)-aconitate, and even the non-physiological cis-homo(4)-aconitate with similar efficiency. These reactions are part of the biosynthesis pathway of coenzyme B. Can also catalyze the hydration of maleate to (R)-malate, and that of cis-aconitate. Can not catalyze the hydration of citraconate and the dehydration of (S)-homocitrate, citramalate, 2-isopropylmalate, 3-isopropylmalate, citrate or threo-DL-isocitrate.<ref>PMID:17449626</ref> <ref>PMID:18765671</ref> <ref>PMID:20170198</ref> | + | [[https://www.uniprot.org/uniprot/HACB_METJA HACB_METJA]] Hydro-lyase with broad substrate specificity for cis-unsaturated tricarboxylic acids. Catalyzes both the reversible dehydration of (R)-homocitrate ((R)-2-hydroxybutane-1,2,4-tricarboxylate) to produce cis-homoaconitate ((Z)-but-1-ene-1,2,4-tricarboxylate), and its hydration to homoisocitrate ((1R,2S)-1-hydroxybutane-1,2,4-tricarboxylate). Is also able to hydrate the analogous longer chain substrates cis-homo(2)-aconitate, cis-homo(3)-aconitate, and even the non-physiological cis-homo(4)-aconitate with similar efficiency. These reactions are part of the biosynthesis pathway of coenzyme B. Can also catalyze the hydration of maleate to (R)-malate, and that of cis-aconitate. Can not catalyze the hydration of citraconate and the dehydration of (S)-homocitrate, citramalate, 2-isopropylmalate, 3-isopropylmalate, citrate or threo-DL-isocitrate.<ref>PMID:17449626</ref> <ref>PMID:18765671</ref> <ref>PMID:20170198</ref> |
| | == Evolutionary Conservation == | | == Evolutionary Conservation == |
| | [[Image:Consurf_key_small.gif|200px|right]] | | [[Image:Consurf_key_small.gif|200px|right]] |
| Line 39: |
Line 39: |
| | __TOC__ | | __TOC__ |
| | </StructureSection> | | </StructureSection> |
| | + | [[Category: Large Structures]] |
| | [[Category: Methanogen homoaconitase]] | | [[Category: Methanogen homoaconitase]] |
| | [[Category: Metja]] | | [[Category: Metja]] |
| Structural highlights
Function
[HACB_METJA] Hydro-lyase with broad substrate specificity for cis-unsaturated tricarboxylic acids. Catalyzes both the reversible dehydration of (R)-homocitrate ((R)-2-hydroxybutane-1,2,4-tricarboxylate) to produce cis-homoaconitate ((Z)-but-1-ene-1,2,4-tricarboxylate), and its hydration to homoisocitrate ((1R,2S)-1-hydroxybutane-1,2,4-tricarboxylate). Is also able to hydrate the analogous longer chain substrates cis-homo(2)-aconitate, cis-homo(3)-aconitate, and even the non-physiological cis-homo(4)-aconitate with similar efficiency. These reactions are part of the biosynthesis pathway of coenzyme B. Can also catalyze the hydration of maleate to (R)-malate, and that of cis-aconitate. Can not catalyze the hydration of citraconate and the dehydration of (S)-homocitrate, citramalate, 2-isopropylmalate, 3-isopropylmalate, citrate or threo-DL-isocitrate.[1] [2] [3]
Evolutionary Conservation
Check, as determined by ConSurfDB. You may read the explanation of the method and the full data available from ConSurf.
Publication Abstract from PubMed
The aconitase family of hydro-lyase enzymes includes three classes of proteins that catalyze the isomerization of alpha-hydroxy acids to beta-hydroxy acids. Besides aconitase, isopropylmalate isomerase (IPMI) proteins specifically catalyze the isomerization of alpha,beta-dicarboxylates with hydrophobic gamma-chain groups, and homoaconitase (HACN) proteins catalyze the isomerization of tricarboxylates with variable chain length gamma-carboxylate groups. These enzymes' stereospecific hydro-lyase activities make them attractive catalysts to produce diastereomers from unsaturated precursors. However, sequence similarity and convergent evolution among these proteins lead to widespread misannotation and uncertainty about gene function. To find the substrate specificity determinants of homologous IPMI and HACN proteins from Methanocaldococcus jannaschii, the small-subunit HACN protein (MJ1271) was crystallized for X-ray diffraction. The structural model showed characteristic residues in a flexible loop region between alpha2 and alpha3 that distinguish HACN from IPMI and aconitase proteins. Site-directed mutagenesis of MJ1271 produced loop-region variant proteins that were reconstituted with wild-type MJ1003 large-subunit protein. The heteromers formed promiscuous hydro-lyases with reduced activity but broader substrate specificity. Both R26K and R26V variants formed relatively efficient IPMI enzymes, while the T27A variant had uniformly lower specificity constants for both IPMI and HACN substrates. The R26V T27Y variant resembles the MJ1277 IPMI small subunit in its flexible loop sequence but demonstrated the broad substrate specificity of the R26V variant. These mutations may reverse the evolution of HACN activity from an ancestral IPMI gene, demonstrating the evolutionary potential for promiscuity in hydro-lyase enzymes. Understanding these specificity determinants enables the functional reannotation of paralogous HACN and IPMI genes in numerous genome sequences. These structural and kinetic results will help to engineer new stereospecific hydro-lyase enzymes for chemoenzymatic syntheses.
Substrate specificity determinants of the methanogen homoaconitase enzyme: structure and function of the small subunit.,Jeyakanthan J, Drevland RM, Gayathri DR, Velmurugan D, Shinkai A, Kuramitsu S, Yokoyama S, Graham DE Biochemistry. 2010 Mar 30;49(12):2687-96. PMID:20170198[4]
From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.
See Also
References
- ↑ Drevland RM, Waheed A, Graham DE. Enzymology and evolution of the pyruvate pathway to 2-oxobutyrate in Methanocaldococcus jannaschii. J Bacteriol. 2007 Jun;189(12):4391-400. Epub 2007 Apr 20. PMID:17449626 doi:http://dx.doi.org/10.1128/JB.00166-07
- ↑ Drevland RM, Jia Y, Palmer DR, Graham DE. Methanogen homoaconitase catalyzes both hydrolyase reactions in coenzyme B biosynthesis. J Biol Chem. 2008 Oct 24;283(43):28888-96. Epub 2008 Sep 2. PMID:18765671 doi:http://dx.doi.org/M802159200
- ↑ Jeyakanthan J, Drevland RM, Gayathri DR, Velmurugan D, Shinkai A, Kuramitsu S, Yokoyama S, Graham DE. Substrate specificity determinants of the methanogen homoaconitase enzyme: structure and function of the small subunit. Biochemistry. 2010 Mar 30;49(12):2687-96. PMID:20170198 doi:10.1021/bi901766z
- ↑ Jeyakanthan J, Drevland RM, Gayathri DR, Velmurugan D, Shinkai A, Kuramitsu S, Yokoyama S, Graham DE. Substrate specificity determinants of the methanogen homoaconitase enzyme: structure and function of the small subunit. Biochemistry. 2010 Mar 30;49(12):2687-96. PMID:20170198 doi:10.1021/bi901766z
|