This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Main Page
From Proteopedia
| Line 45: | Line 45: | ||
<td style="padding: 10px;> | <td style="padding: 10px;> | ||
| - | <p>[[I3DC|About Interactive 3D | + | <p>[[I3DC|About Interactive 3D Complements ('''I3DCs''')]]</p> |
| - | <p>[[Proteopedia:I3DC|List of | + | <p>[[Proteopedia:I3DC|List of I3DCs]]</p> |
| - | <p>[[How to get an I3DC | + | <p>[[How to get an I3DC for your paper]]</p> |
</td> | </td> | ||
Revision as of 09:53, 21 October 2018
|
ISSN 2310-6301
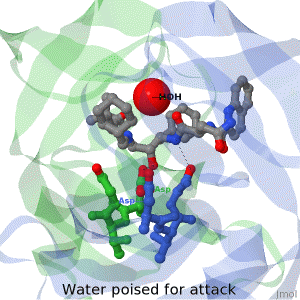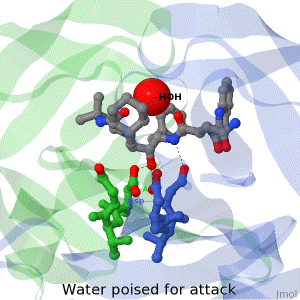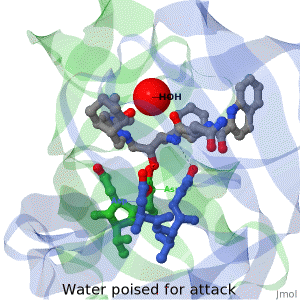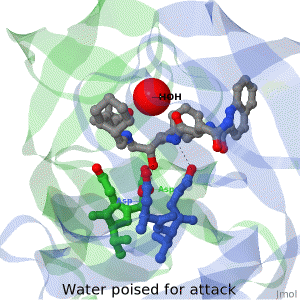
As life is more than 2D, Proteopedia helps to bridge the 3D relationships between function & structure of biomacromolecules
| |||||||||||
| Selected Pages | Art on Science | Journals | Education | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
||||||||
|
How to add content to Proteopedia Who knows ... |
Teaching Strategies Using Proteopedia |
||||||||||
| |||||||||||