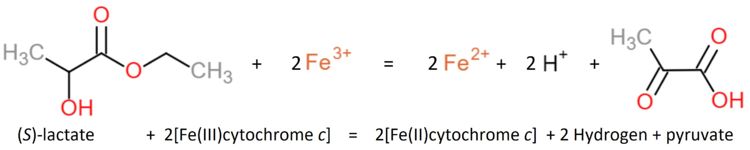This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1501
From Proteopedia
(Difference between revisions)
| Line 18: | Line 18: | ||
<!-- Global Stoichiometry Homotetramer A4 --> | <!-- Global Stoichiometry Homotetramer A4 --> | ||
| - | Flavocytochrome b(2) is a tetrameric enzyme (Jacq and Lederer, 1972 and 1974<ref>1972PMID: 4336855</ref>,<ref>1972PMID: 4152980</ref> | + | Flavocytochrome b(2) is a tetrameric enzyme (Jacq and Lederer, 1972 and 1974<ref>1972PMID: 4336855</ref>,<ref>1972PMID: 4152980</ref>). Each of the four identical subunits is composed by one single polypeptide chain. |
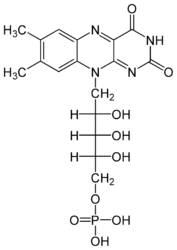
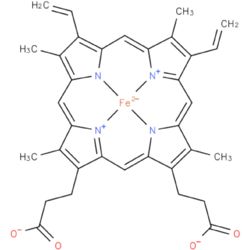
| - | Each subunit contains a binding site for the selectively non-covalently binding of the cofactor FMN(3-) (Flavinmononucleotide), as well as one in with the iron complexed in the tetrapyrrole ring interacts with heme b(2-) cofactor (Risler and Groudinsky, 1973 | + | Each subunit contains a binding site for the selectively non-covalently binding of the cofactor FMN(3-) (Flavinmononucleotide), as well as one in with the iron complexed in the tetrapyrrole ring interacts with heme b(2-) cofactor (Risler and Groudinsky, 1973<ref>PMID: 5545004 </ref>). |
| + | <!--https://www.wikiwand.com/de/Flavinmononukleotid--> | ||
<!-- https://www.ebi.ac.uk/chebi/searchId.do?chebiId=CHEBI:60344--> | <!-- https://www.ebi.ac.uk/chebi/searchId.do?chebiId=CHEBI:60344--> | ||
Revision as of 18:52, 10 January 2019
| This Sandbox is Reserved from 06/12/2018, through 30/06/2019 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1480 through Sandbox Reserved 1543. |
To get started:
More help: Help:Editing |
| |||||||||||
References
- ↑ NYGAARD AP. Various forms of D- and L-lactate dehydrogenases in yeast. Ann N Y Acad Sci. 1961 Nov 2;94:774-9. PMID:14480786
- ↑ https://doi.org/10.1016/0076-6879(66)09064-5
- ↑ NYGAARD AP. Various forms of D- and L-lactate dehydrogenases in yeast. Ann N Y Acad Sci. 1961 Nov 2;94:774-9. PMID:14480786
- ↑ 1972PMID: 4336855
- ↑ 1972PMID: 4152980
- ↑ Groudinsky O. Study of heme-protein linkage in cytochrome b2. Destruction of a crucial histidine residue by photooxidation of "apo" cytochrome b2 core in the presence of rose bengal. Eur J Biochem. 1971 Feb;18(4):480-4. PMID:5545004
- ↑ 1972PMID: 4575975
- ↑ APPLEBY CA, MORTON RK. Lactic dehydrogenase and cytochrome b2 of baker's yeast; purification and crystallization. Biochem J. 1959 Mar;71(3):492-9. PMID:13638255
- ↑ Lederer F. On the first steps of lactate oxidation by bakers' yeast L-(plus)-lactate dehydrogenase (cytochrome b2). Eur J Biochem. 1974 Jul 15;46(2):393-9. PMID:4152980
- ↑ Tegoni M, Cambillau C. The 2.6-A refined structure of the Escherichia coli recombinant Saccharomyces cerevisiae flavocytochrome b2-sulfite complex. Protein Sci. 1994 Feb;3(2):303-13. PMID:8003966
- ↑ Tegoni M, Cambillau C. The 2.6-A refined structure of the Escherichia coli recombinant Saccharomyces cerevisiae flavocytochrome b2-sulfite complex. Protein Sci. 1994 Feb;3(2):303-13. PMID:8003966