This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Citrate Synthase
From Proteopedia
(Difference between revisions)
| Line 38: | Line 38: | ||
*<scene name='Citrate_Synthase/Cv/1'>Citrate Synthase Closed Form (Monomer)</scene> [[2cts]] | *<scene name='Citrate_Synthase/Cv/1'>Citrate Synthase Closed Form (Monomer)</scene> [[2cts]] | ||
*<scene name='Citrate_Synthase/Cv/2'>'Citrate Synthase Open Form (Monomer)</scene> [[1cts]] | *<scene name='Citrate_Synthase/Cv/2'>'Citrate Synthase Open Form (Monomer)</scene> [[1cts]] | ||
| + | |||
| + | ==3D structures of Citrate Synthase== | ||
| + | [[Citrate Synthase 3D structures]] | ||
| + | |||
</StructureSection> | </StructureSection> | ||
__NOTOC__ | __NOTOC__ | ||
| Line 51: | Line 55: | ||
*Citrate synthase | *Citrate synthase | ||
| - | **[[ | + | **[[5uzq]], [[5uzr]] - hCitS – human<br /> |
**[[3enj]] – CitS – Wild boar<br /> | **[[3enj]] – CitS – Wild boar<br /> | ||
| + | **[[1cts]], [[2cts]] – pCitS - pig<br /> | ||
| + | **[[5csc]], [[3cts]] – cCitS – chicken<br /> | ||
**[[2p2w]] – CitS – ''Thermotoga maritima''<br /> | **[[2p2w]] – CitS – ''Thermotoga maritima''<br /> | ||
**[[2c6x]] – CitS residues 2-364 – ''Bacillus subtilis''<br /> | **[[2c6x]] – CitS residues 2-364 – ''Bacillus subtilis''<br /> | ||
| Line 62: | Line 68: | ||
**[[1a59]] – CitS – ''Antarctic bacterium''<br /> | **[[1a59]] – CitS – ''Antarctic bacterium''<br /> | ||
**[[1aj8]] – CitS - ''Pyrococcus furiosus''<br /> | **[[1aj8]] – CitS - ''Pyrococcus furiosus''<br /> | ||
| - | **[[5csc]], [[3cts]]– cCitS – chicken<br /> | ||
| - | **[[1cts]], [[2cts]] – pCitS - pig<br /> | ||
**[[4e6y]] – CitS – ''Vibrio vulnificus''<br /> | **[[4e6y]] – CitS – ''Vibrio vulnificus''<br /> | ||
**[[4tvm]] – MtCitS – ''Mycobacterium tuberculosis''<br /> | **[[4tvm]] – MtCitS – ''Mycobacterium tuberculosis''<br /> | ||
**[[4xgh]] – CitS – ''Burkholderia thailandensis''<br /> | **[[4xgh]] – CitS – ''Burkholderia thailandensis''<br /> | ||
**[[4ybo]] - TaCitS – ''Thermoplasma acidophilum''<br /> | **[[4ybo]] - TaCitS – ''Thermoplasma acidophilum''<br /> | ||
| - | **[[ | + | **[[3msu]] – CitS – ''Francisella tularensis''<br /> |
| + | **[[6abv]] – MsCitS – ''Metallosphaera sedula''<br /> | ||
*Citrate synthase binary complex | *Citrate synthase binary complex | ||
| - | **[[ | + | **[[5uzp]] - hCitS (mutant) + oxaloacetate <br /> |
| - | + | ||
**[[6cts]] – cCitS + citryl thioether CoA<br /> | **[[6cts]] – cCitS + citryl thioether CoA<br /> | ||
| + | **[[4jae]] - EcCitS (mutant) + S-carboxymethyl-CoA<br /> | ||
**[[1owb]], [[1nxg]], [[4jaf]] - EcCitS (mutant) + NADH<br /> | **[[1owb]], [[1nxg]], [[4jaf]] - EcCitS (mutant) + NADH<br /> | ||
**[[4jag]] - EcCitS (mutant) + oxaloacetate<br /> | **[[4jag]] - EcCitS (mutant) + oxaloacetate<br /> | ||
**[[2ifc]] - TaCitS + oxaloacetate<br /> | **[[2ifc]] - TaCitS + oxaloacetate<br /> | ||
| - | **[[ | + | **[[2r9e]] – TaCitS + citryl dethia CoA <br /> |
| + | **[[6abw]] - MsCitS + acetyl CoA<br /> | ||
| + | **[[6abx]] - MsCitS + citrate<br /> | ||
| + | **[[6aby]] - MsCitS + oxaloacetate <br /> | ||
*Citrate synthase ternary complex | *Citrate synthase ternary complex | ||
| - | **[[ | + | **[[4cts]] - pCitS + oxaloacetate + S-acetonyl CoA<br /> |
| - | + | ||
**[[1al6]], [[1csr]], [[1css]], [[1csh]], [[1csi]] - cCitS + oxaloacetate + carboxymethyl dethia-CoA derivative<br /> | **[[1al6]], [[1csr]], [[1css]], [[1csh]], [[1csi]] - cCitS + oxaloacetate + carboxymethyl dethia-CoA derivative<br /> | ||
**[[5cts]], [[6cts]] - cCitS + oxaloacetate + carboxymethyl CoA<br /> | **[[5cts]], [[6cts]] - cCitS + oxaloacetate + carboxymethyl CoA<br /> | ||
| Line 89: | Line 96: | ||
**[[1csc]], [[2csc]], [[3csc]], [[4csc]] - cCitS + malate + carboxymethyl CoA<br /> | **[[1csc]], [[2csc]], [[3csc]], [[4csc]] - cCitS + malate + carboxymethyl CoA<br /> | ||
**[[6csc]] - cCitS + citrate + trifluoroacetonyl-CoA<br /> | **[[6csc]] - cCitS + citrate + trifluoroacetonyl-CoA<br /> | ||
| - | **[[ | + | **[[2r26]] - TaCitS + oxaloacetate + S-carboxymethyl-CoA<br /> |
| + | **[[2h12]] - CitS + oxaloacetate + carboxymethyl dethia-CoA – ''Acetobacter aceti''<br /> | ||
* ATP-Citrate synthase see [[ATP-citrate synthase]] | * ATP-Citrate synthase see [[ATP-citrate synthase]] | ||
| Line 98: | Line 106: | ||
**[[3tqg]] – 2-MCitS – ''Coxiella burnetii''<br /> | **[[3tqg]] – 2-MCitS – ''Coxiella burnetii''<br /> | ||
**[[3o8j]] – 2-MCitS – ''Salmonella enterica''<br /> | **[[3o8j]] – 2-MCitS – ''Salmonella enterica''<br /> | ||
| - | **[[5uqo]], [[5uqq]], [[5uqs]] – 2-AfMCitS – ''Aspergillus fumigata''<br /> | + | **[[5uqo]], [[5uqq]], [[5uqs]], [[6bop]] – 2-AfMCitS – ''Aspergillus fumigata''<br /> |
| - | **[[ | + | **[[6bol]], [[6bom]] – 2-AfMCitS (mutant) + oxaloacetate <br /> |
| + | **[[5uqr]] – 2-AfMCitS + oxaloacetate + ethyl CoA<br /> | ||
| + | **[[6bon]], [[6boo]] – 2-AfMCitS + oxaloacetate + CoA<br /> | ||
**[[5uqu]] – 2-AfMCitS (mutant) + oxaloacetate + CoA<br /> | **[[5uqu]] – 2-AfMCitS (mutant) + oxaloacetate + CoA<br /> | ||
}} | }} | ||
Revision as of 09:57, 13 May 2019
This page, as it appeared on March 3, 2011, was featured in this article in the journal Biochemistry and Molecular Biology Education.
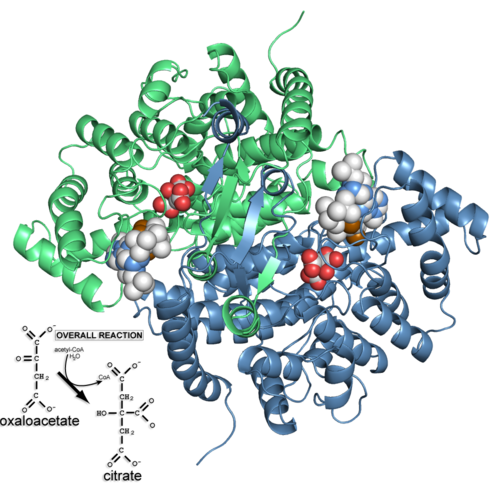
Citrate synthase 'closed' form complex with CoA and citrate (2cts) and the reaction
3D structures of Citrate Synthase
Updated on 13-May-2019
See Also
Literature and Notes
- ↑ "Citrate Synthase -." Wikipedia, the Free Encyclopedia. Web. 22 Mar. 2010.
- ↑ 2.0 2.1 Voet, Donald, Judith G. Voet, and Charlotte W. Pratt. Fundamentals of Biochemistry: Life at the Molecular Level. Hoboken, NJ: Wiley, 2008.
- ↑ 3.0 3.1 3.2 Remington S, Wiegand G, Huber R. Crystallographic refinement and atomic models of two different forms of citrate synthase at 2.7 and 1.7 A resolution. J Mol Biol. 1982 Jun 15;158(1):111-52. PMID:7120407
- ↑ In this structure 1cts, citrate, the resulting product of the conversion, is actually bound where oxaloacetate binds.
- ↑ Bayer E, Bauer B, Eggerer H. Evidence from inhibitor studies for conformational changes of citrate synthase. Eur J Biochem. 1981 Nov;120(1):155-60. PMID:7308213
- ↑ Karpusas M, Branchaud B, Remington SJ. Proposed mechanism for the condensation reaction of citrate synthase: 1.9-A structure of the ternary complex with oxaloacetate and carboxymethyl coenzyme A. Biochemistry. 1990 Mar 6;29(9):2213-9. PMID:2337600
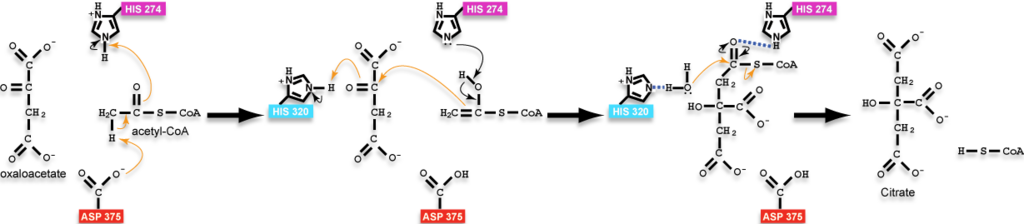
- ↑ 5cts as the state preceding condensation with oxaloacetate and a non-reactive version of acetyl-CoA bound, 6cts as the state containing the bound intermediate, and 3cts as the complex with the products. Positions of hydrogens on the ligands were calculated and added back to structures in the reaction scheme for instructional purposes and are not present in the experimentally-determined structures; additionally, arrows are drawn with atoms of the analog of acetyl-CoA to approximate the position of the reactive groups only as the reactive groups are not actually part of the analog or the molecules would have reacted; please, see the reaction scheme on this page for a more thorough accounting of the chemistry.
- ↑ Wiegand G, Remington SJ. Citrate synthase: structure, control, and mechanism. Annu Rev Biophys Biophys Chem. 1986;15:97-117. PMID:3013232 doi:http://dx.doi.org/10.1146/annurev.bb.15.060186.000525
- ↑ Kim KS, Rosenkrantz MS, Guarente L. Saccharomyces cerevisiae contains two functional citrate synthase genes. Mol Cell Biol. 1986 Jun;6(6):1936-42. PMID:3023912
- ↑ Lewin AS, Hines V, Small GM. Citrate synthase encoded by the CIT2 gene of Saccharomyces cerevisiae is peroxisomal. Mol Cell Biol. 1990 Apr;10(4):1399-405. PMID:2181273
- ↑ Lee YJ, Hoe KL, Maeng PJ. Yeast cells lacking the CIT1-encoded mitochondrial citrate synthase are hypersusceptible to heat- or aging-induced apoptosis. Mol Biol Cell. 2007 Sep;18(9):3556-67. Epub 2007 Jul 5. PMID:17615299 doi:10.1091/mbc.E07-02-0118
External Resources
- Citrate Synthase: September 2007 Molecule of the Month as part of the series of tutorials that are at the RCSB Protein Data Bank and written by David Goodsell
- An interactive schematic animation of Citrate synthase's reaction mechanism from Lehninger's Principles of Biochemistry
- Citrate Synthase at Wikipedia
Proteopedia Page Contributors and Editors (what is this?)
Wayne Decatur, Michal Harel, Daniel Eddelman, Alexander Berchansky, Joel L. Sussman, Angel Herraez, David Canner, Eric Martz