This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Bacterial thiolase
From Proteopedia
(Difference between revisions)
| Line 78: | Line 78: | ||
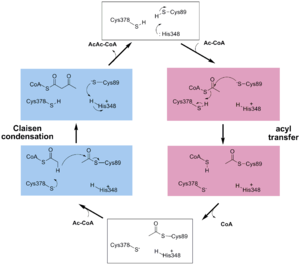
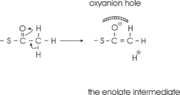
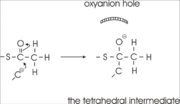
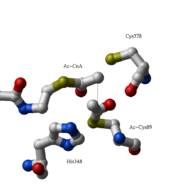
site geometry: a Asn316-Wat82 dyad, together with His348 make <scene name='User:Satyan_Sharma/Sandbox_1/Oxyanionhole_1/2'>oxyanion hole 1</scene>, stabilizing the CoA-thioester enolate intermediate. The hydrogen bond donors of oxyanion hole 1 are Wat82 and NE2 (His348). This enolate is formed from acetyl-CoA after proton | site geometry: a Asn316-Wat82 dyad, together with His348 make <scene name='User:Satyan_Sharma/Sandbox_1/Oxyanionhole_1/2'>oxyanion hole 1</scene>, stabilizing the CoA-thioester enolate intermediate. The hydrogen bond donors of oxyanion hole 1 are Wat82 and NE2 (His348). This enolate is formed from acetyl-CoA after proton | ||
abstraction by the catalytic base, Cys378 (Figure 3). | abstraction by the catalytic base, Cys378 (Figure 3). | ||
| - | <applet load='1dm3' size='300' frame='true' align='right' caption='The bacterial biosynthetic thiolase showing the acetyl CoA complex with sulfate: [[1DM3]], resolution 2.00 Å.'/> | ||
| Line 143: | Line 142: | ||
''Zoogloea ramigera'' is indeed important for the function of oxyanion | ''Zoogloea ramigera'' is indeed important for the function of oxyanion | ||
hole 1. | hole 1. | ||
| - | </StructureSection | + | </StructureSection> |
==Additional Resources== | ==Additional Resources== | ||
For additional information, see: [[Metabolic Disorders]] | For additional information, see: [[Metabolic Disorders]] | ||
Revision as of 09:47, 4 February 2019
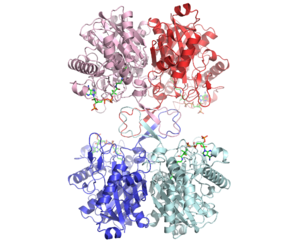
3D structure (1DM3) of the bacterial Zoogloea ramigera biosynthetic thiolase
| |||||||||||
Additional Resources
For additional information, see: Metabolic Disorders
3D structures of Thiolase
References
- ↑ Williams SF, Palmer MA, Peoples OP, Walsh CT, Sinskey AJ, Masamune S. Biosynthetic thiolase from Zoogloea ramigera. Mutagenesis of the putative active-site base Cys-378 to Ser-378 changes the partitioning of the acetyl S-enzyme intermediate. J Biol Chem. 1992 Aug 15;267(23):16041-3. PMID:1353760
- ↑ 2.0 2.1 Kursula P, Ojala J, Lambeir AM, Wierenga RK. The catalytic cycle of biosynthetic thiolase: a conformational journey of an acetyl group through four binding modes and two oxyanion holes. Biochemistry. 2002 Dec 31;41(52):15543-56. PMID:12501183
- ↑ 3.0 3.1 3.2 Merilainen G, Poikela VM, Kursula P, Wierenga RK. The thiolase reaction mechanism: the importance of Asn316 and His348 for stabilizing the enolate intermediate of the Claisen condensation. Biochemistry. 2009 Oct 20. PMID:19842716 doi:10.1021/bi901069h
- ↑ 4.0 4.1 Fukao T, Nguyen HT, Nguyen NT, Vu DC, Can NT, Pham AT, Nguyen KN, Kobayashi H, Hasegawa Y, Bui TP, Niezen-Koning KE, Wanders RJ, de Koning T, Nguyen LT, Yamaguchi S, Kondo N. A common mutation, R208X, identified in Vietnamese patients with mitochondrial acetoacetyl-CoA thiolase (T2) deficiency. Mol Genet Metab. 2010 May;100(1):37-41. Epub 2010 Jan 21. PMID:20156697 doi:10.1016/j.ymgme.2010.01.007
- ↑ Modis Y, Wierenga RK. Crystallographic analysis of the reaction pathway of Zoogloea ramigera biosynthetic thiolase. J Mol Biol. 2000 Apr 14;297(5):1171-82. PMID:10764581 doi:10.1006/jmbi.2000.3638
- ↑ Haapalainen AM, Merilainen G, Wierenga RK. The thiolase superfamily: condensing enzymes with diverse reaction specificities. Trends Biochem Sci. 2006 Jan;31(1):64-71. Epub 2005 Dec 13. PMID:16356722 doi:10.1016/j.tibs.2005.11.011
- ↑ Jiang C, Kim SY, Suh DY. Divergent evolution of the thiolase superfamily and chalcone synthase family. Mol Phylogenet Evol. 2008 Dec;49(3):691-701. Epub 2008 Sep 12. PMID:18824113 doi:10.1016/j.ympev.2008.09.002
Proteopedia Page Contributors and Editors (what is this?)
Rik Wierenga, Joel L. Sussman, Michal Harel, Satyan Sharma, David Canner