This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Jordan Finch/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
| - | <Structure load='4psw' size='350' frame='true' align='right' caption='Hat1/Hat2 Complex' scene='81/811713/4psw_overview/1'></scene> | ||
| - | |||
==Histone Acetyltransferase HAT1/HAT2 Complex, ''Saccharomyces cerevisiae''== | ==Histone Acetyltransferase HAT1/HAT2 Complex, ''Saccharomyces cerevisiae''== | ||
| + | <StructureSection load='4psw' size='350' frame='true' side='right' caption='HAT1 4PSW' | ||
| + | This is a default text for your page '''Caitlin Marie Gaich/Sandbox1'''. Click above on '''edit this page''' to modify. Be careful with the < and > signs. | ||
| + | You may include any references to papers as in: the use of JSmol in Proteopedia <ref>DOI 10.1002/ijch.201300024</ref> or to the article describing Jmol <ref>PMID:21638687</ref> to the rescue. | ||
== Introduction == | == Introduction == | ||
| Line 9: | Line 10: | ||
[[Image:Main_bonding_cartoon_3.png|400px|right|thumb|Figure 1]] | [[Image:Main_bonding_cartoon_3.png|400px|right|thumb|Figure 1]] | ||
== Hat1/Hat2 Complex Structure == | == Hat1/Hat2 Complex Structure == | ||
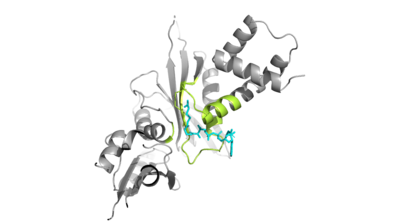
| - | + | Hat1 is not catalytically active until it binds with HAT2 to form the <scene name='81/811718/Hat1_hat2_complex_aco_and_h4/1'>complex</scene>. HAT1 structure includes 317 residues and is identified as <scene name='81/811718/Hat1_protein/1'>chain A</scene>. HAT2 is identified as <scene name='81/811718/Hat2_protein/1'>chain B</scene>, which includes 401 residues. The activated complex acetylates residues in the 38 residue span of <scene name='81/811718/Portion_of_histone_4/1'>Histone 4</scene>. | |
| + | |||
== Mechanism == | == Mechanism == | ||
| Line 15: | Line 17: | ||
In this mechanism, the glutamate at residue 255 acts as a general base and deprotonates lysine 12 of histone 4 (the numbering of the modified lysine residue on histone 4 is shifted two residues).The deprotonated lysine then acts as a nucleophile and attacks the carbonyl carbon of acetyl coenzyme A (CoA, forming a tetrahedral intermediate transition state. The negative charge on the oxygen collapses down to for a double bond and the scissle bond between the carbonyl carbon and the sulfur atom of acetyl CoA is broken. The resulting product of this reaction is histone 4 with an acetyl-lysine at residue 12 and CoEnzyme A. | In this mechanism, the glutamate at residue 255 acts as a general base and deprotonates lysine 12 of histone 4 (the numbering of the modified lysine residue on histone 4 is shifted two residues).The deprotonated lysine then acts as a nucleophile and attacks the carbonyl carbon of acetyl coenzyme A (CoA, forming a tetrahedral intermediate transition state. The negative charge on the oxygen collapses down to for a double bond and the scissle bond between the carbonyl carbon and the sulfur atom of acetyl CoA is broken. The resulting product of this reaction is histone 4 with an acetyl-lysine at residue 12 and CoEnzyme A. | ||
| + | |||
== Application == | == Application == | ||
== References == | == References == | ||
| - | + | Li, Y. et. al. ''Hat2p recognizes the histone H3 tail to specify the acetylation of the newly synthesized H3/H4 heterodimer by the Hat1p/Hat2p complex.''(2014). ''Genes Dev.''28:1217-1227. DOI:10.1101/gad.240531.114 | |
<references/> | <references/> | ||
Revision as of 18:10, 5 April 2019
Histone Acetyltransferase HAT1/HAT2 Complex, Saccharomyces cerevisiae
| |||||||||||