This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Courtney Brown/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 43: | Line 43: | ||
Substrates are deacetylated via a classic 2<sup>+</sup> metal ion mechanism. Zinc pentacoordinates with <scene name='81/811715/Residues_coordinating_to_zinc/1'>Asp178, His180, Asp267</scene>, the nucleophilic water and the carbonyl oxygen of the substrate. | Substrates are deacetylated via a classic 2<sup>+</sup> metal ion mechanism. Zinc pentacoordinates with <scene name='81/811715/Residues_coordinating_to_zinc/1'>Asp178, His180, Asp267</scene>, the nucleophilic water and the carbonyl oxygen of the substrate. | ||
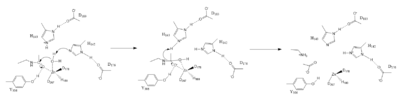
| - | The Zinc ion <scene name='81/811715/Zn_activating_water/1'>activates the water</scene> by withdrawing electron density from the water due to the positive charge, making the water more acidic, therefore a better nucleophile. The Zinc ion also coordinates to the carbonyl oxygen of the acetyl group on the lysine, polarizing the carbonyl carbon, making it more electrophilic. <scene name='81/811715/His_142_and_143/1'>His142</scene> deprotinates the water, the first step of the deacetylation (Figure 1). His 142 is closer to the water molecule than His 143, which was also thought to deprotinate the water. However, a mutation done to His 143 only reduced activity, not abolished it, showing it is important but not crucial. His 143 is thought to orient the substrate | + | The Zinc ion <scene name='81/811715/Zn_activating_water/1'>activates the water</scene> by withdrawing electron density from the water due to the positive charge, making the water more acidic, therefore a better nucleophile. The Zinc ion also coordinates to the carbonyl oxygen of the acetyl group on the lysine, polarizing the carbonyl carbon, making it more electrophilic. <scene name='81/811715/His_142_and_143/1'>His142</scene> deprotinates the water, the first step of the deacetylation (Figure 1). His 142 (stabilized by Asp 176) is closer to the water molecule than His 143 (stabilized by Asp 183), which was also thought to deprotinate the water. However, a mutation done to His 143 only reduced activity, not abolished it, showing it is important but not crucial. His 143 is instead thought to orient the substrate<ref name="Vanninni" />. |
| - | The attack by the deprotinated water forces the carbonyl carbon into a tetrehedral transition state. <scene name='81/811715/Tyr306/4'>Tyr306</scene> acts as the oxyanion hole, stabilizing this transition state via its protruding -OH group hydrogen-bonding with the negatively charged oxygen. | + | The attack by the deprotinated water forces the carbonyl carbon into a tetrehedral transition state. <scene name='81/811715/Tyr306/4'>Tyr306</scene> acts as the oxyanion hole, stabilizing this transition state via its protruding -OH group hydrogen-bonding with the negatively charged oxygen. The transition state collapses, and His 143 simultaneously gets deprotinated by the amide of the leaving acetyl group in order to weaken the scissle bond. Breaking this scissle bond results in a neutral lysine and a carboxylic acid, among other products. Body pH is around 7 typically and the pKa of carboxylic acids is around 2; the hydrogen from the COOH group is lost and picked up by the lysine, providing the hydrogen necessary for lysine to become acidic, per its nature. |
==Disease== | ==Disease== | ||
Revision as of 03:00, 7 April 2019
The Human Histone H3/K9 Deacetylase, HDAC8
| |||||||||||