This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Jordan Finch/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 11: | Line 11: | ||
== Hat1/Hat2 Complex Structure == | == Hat1/Hat2 Complex Structure == | ||
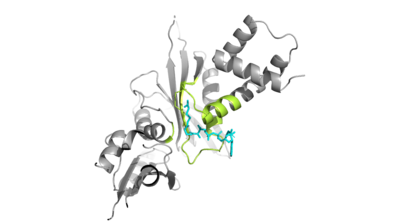
Hat1 is not catalytically active until it binds with HAT2 to form the <scene name='81/811718/Hat1_hat2_complex_aco_and_h4/1'>complex</scene>. HAT1 structure includes 317 residues and is identified as <scene name='81/811718/Hat1_protein/1'>chain A</scene>. HAT2 is identified as <scene name='81/811718/Hat2_protein/1'>chain B</scene>, which includes 401 residues. The activated complex acetylates residues in the 38 residue span of <scene name='81/811718/Portion_of_histone_4/1'>Histone 4</scene>. | Hat1 is not catalytically active until it binds with HAT2 to form the <scene name='81/811718/Hat1_hat2_complex_aco_and_h4/1'>complex</scene>. HAT1 structure includes 317 residues and is identified as <scene name='81/811718/Hat1_protein/1'>chain A</scene>. HAT2 is identified as <scene name='81/811718/Hat2_protein/1'>chain B</scene>, which includes 401 residues. The activated complex acetylates residues in the 38 residue span of <scene name='81/811718/Portion_of_histone_4/1'>Histone 4</scene>. | ||
| - | The HAT1 and HAT2 interface is stabilized by hydrogen bonds, salt bridges, and hydrophobic interactions. Most of these interactions are located in LP1 of the HAT1 domain, which forms a well-ordered helix. There are three major areas where hydrogen bonds are present. The side chain atoms of <scene name='81/811717/Lys211phe205_and_leu288arg282/5'>Tyr199 and Asp308</scene> with the main chain nitrogen of Ala202 in HAT1. The side chain of <scene name='81/811717/Lys211phe205_and_leu288arg282/4'>Lys211 and Arg282</scene> makes hydrogen bonds with Leu288 and Phe205 respectively. The last area of hydrogen bonds between HAT1 and | + | The HAT1 and HAT2 interface is stabilized by hydrogen bonds, salt bridges, and hydrophobic interactions. Most of these interactions are located in LP1 of the HAT1 domain, which forms a well-ordered helix. There are three major areas where hydrogen bonds are present. The side chain atoms of <scene name='81/811717/Lys211phe205_and_leu288arg282/5'>Tyr199 and Asp308</scene> with the main chain nitrogen of Ala202 in HAT1. The side chain of <scene name='81/811717/Lys211phe205_and_leu288arg282/4'>Lys211 and Arg282</scene> makes hydrogen bonds with Leu288 and Phe205 respectively. The last area of hydrogen bonds between HAT1 and HAT2 is found between <scene name='81/811717/Serine_hydrogen_bonds/2'>Ser263 and Asp 206</scene>. |
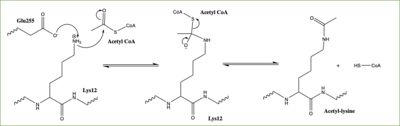
== Mechanism == | == Mechanism == | ||
Revision as of 20:24, 10 April 2019
Histone Acetyltransferase HAT1/HAT2 Complex, Saccharomyces cerevisiae
| |||||||||||