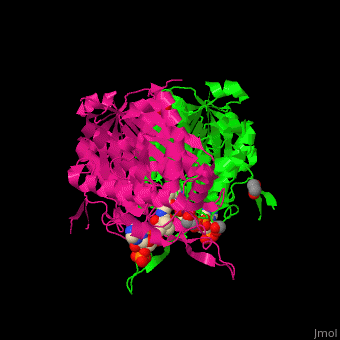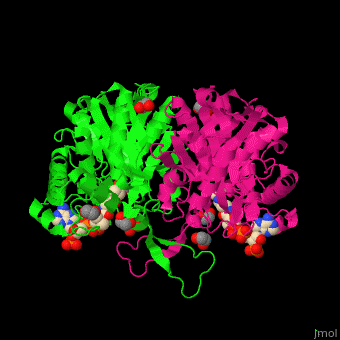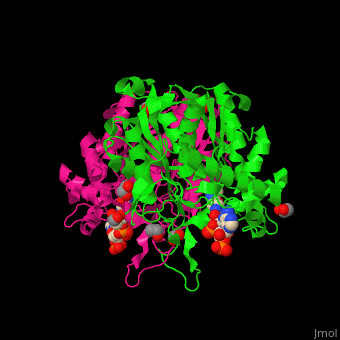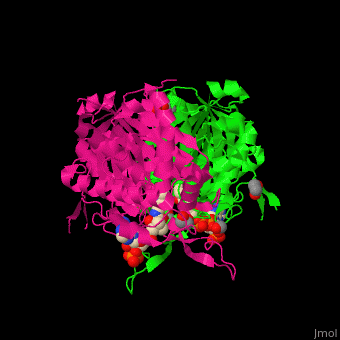This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Thiolase
From Proteopedia
(Difference between revisions)
| Line 11: | Line 11: | ||
== Structural highlights == | == Structural highlights == | ||
The <scene name='44/447919/Cv/8'>active site of 3-ketoacyl-CoA thiolase contains CoA</scene><ref>PMID:25478839</ref>. Water molecules are shown as red spheres. | The <scene name='44/447919/Cv/8'>active site of 3-ketoacyl-CoA thiolase contains CoA</scene><ref>PMID:25478839</ref>. Water molecules are shown as red spheres. | ||
| + | == 3D Structures of Thiolase == | ||
| + | [[Thiolase 3D structures]] | ||
| + | |||
</StructureSection> | </StructureSection> | ||
== 3D Structures of Thiolase == | == 3D Structures of Thiolase == | ||
| Line 19: | Line 22: | ||
*3-ketoacyl-CoA thiolase | *3-ketoacyl-CoA thiolase | ||
| - | **[[2wu9]], [[2c7y]], [[2c7z]] – KCT peroxisomal residues 36-462 – ''Arabidopsis thaliana''<br /> | ||
| - | **[[2wua]] - KCT peroxisomal residues 17-449 (mutant) – sunflower<br /> | ||
**[[2iik]] – hKCT peroxisomal – human<br /> | **[[2iik]] – hKCT peroxisomal – human<br /> | ||
**[[4c2k]] – hKCT mitochondrial <br /> | **[[4c2k]] – hKCT mitochondrial <br /> | ||
**[[4c2j]] – hKCT mitochondrial + CoA<br /> | **[[4c2j]] – hKCT mitochondrial + CoA<br /> | ||
| + | **[[2wu9]], [[2c7y]], [[2c7z]] – KCT peroxisomal residues 36-462 – ''Arabidopsis thaliana''<br /> | ||
| + | **[[2wua]] - KCT peroxisomal residues 17-449 (mutant) – sunflower<br /> | ||
| + | **[[1afw]], [[1pxt]] – yKCT peroxisomal – yeast<br /> | ||
**[[3goa]] – KCT – ''Salmonella typhimurium''<br /> | **[[3goa]] – KCT – ''Salmonella typhimurium''<br /> | ||
**[[5lp7]] – KCT – ''Bacillus subtilis'' <br /> | **[[5lp7]] – KCT – ''Bacillus subtilis'' <br /> | ||
| - | **[[1afw]], [[1pxt]] – yKCT peroxisomal – yeast<br /> | ||
**[[5cbq]] – MsKCT – ''Mycobacterium smegmatis'' <br /> | **[[5cbq]] – MsKCT – ''Mycobacterium smegmatis'' <br /> | ||
**[[4w61]] – ReKCT – ''Ralstonia eutropha''<br /> | **[[4w61]] – ReKCT – ''Ralstonia eutropha''<br /> | ||
| Line 43: | Line 46: | ||
*Acetoacetyl-CoA thiolase or acetyl-CoA acetyltransferase | *Acetoacetyl-CoA thiolase or acetyl-CoA acetyltransferase | ||
| - | **[[1m4s]], [[1m4t]], [[1dlu]], [[1qfl]] - ZrACT – ''Zoogloea ramigera''<br /> | ||
| - | **[[2wku]], [[2wl6]], [[1m1t]], [[1m3k]] - ZrACT (mutant) <br /> | ||
**[[2ib7]], [[2ib8]], [[2ib9]], [[2ibu]], [[2ibw]], [[2iby]], [[2f2s]] – hACT mitochondrial<br /> | **[[2ib7]], [[2ib8]], [[2ib9]], [[2ibu]], [[2ibw]], [[2iby]], [[2f2s]] – hACT mitochondrial<br /> | ||
**[[1wl5]] - hACT cytosolic<br /> | **[[1wl5]] - hACT cytosolic<br /> | ||
| + | **[[5xyj]], [[5xz5]] – yACT <br /> | ||
| + | **[[1m4s]], [[1m4t]], [[1dlu]], [[1qfl]] - ZrACT – ''Zoogloea ramigera''<br /> | ||
| + | **[[2wku]], [[2wl6]], [[1m1t]], [[1m3k]] - ZrACT (mutant) <br /> | ||
**[[4dd5]], [[4e1l]] – ACT – ''Clostridium difficile''<br /> | **[[4dd5]], [[4e1l]] – ACT – ''Clostridium difficile''<br /> | ||
**[[4xl2]], [[4xl3]], [[4n44]], [[4n45]] – CaACT – ''Clostridium acetobutylicum''<br /> | **[[4xl2]], [[4xl3]], [[4n44]], [[4n45]] – CaACT – ''Clostridium acetobutylicum''<br /> | ||
| Line 52: | Line 56: | ||
**[[5f0v]], [[5f38]], [[4wys]] – ACT – ''Escherichia coli'' <br /> | **[[5f0v]], [[5f38]], [[4wys]] – ACT – ''Escherichia coli'' <br /> | ||
**[[4o9a]], [[4o99]], [[4o9c]], [[4nzs]] – ReACT <br /> | **[[4o9a]], [[4o99]], [[4o9c]], [[4nzs]] – ReACT <br /> | ||
| - | **[[5xyj]], [[5xz5]] – yACT <br /> | ||
*Acetoacetyl-CoA thiolase complex | *Acetoacetyl-CoA thiolase complex | ||
| + | **[[1wl4]] – hACT cytosolic + CoA <br /> | ||
**[[1dm3]], [[1dlv]] – ZrACT + CoA<br /> | **[[1dm3]], [[1dlv]] – ZrACT + CoA<br /> | ||
**[[2wkt]], [[2wkv]], [[2wl4]], [[2wl5]], [[2vtz]], [[1m3z]] – ZrACT (mutant) + CoA<br /> | **[[2wkt]], [[2wkv]], [[2wl4]], [[2wl5]], [[2vtz]], [[1m3z]] – ZrACT (mutant) + CoA<br /> | ||
| Line 61: | Line 65: | ||
**[[2vu0]], [[1nl7]] - ZrACT + CoA<br /> | **[[2vu0]], [[1nl7]] - ZrACT + CoA<br /> | ||
**[[2vu1]], [[2vu2]], [[1ou6]] - ZrACT + pantheteine-11-pivalate<br /> | **[[2vu1]], [[2vu2]], [[1ou6]] - ZrACT + pantheteine-11-pivalate<br /> | ||
| - | **[[1wl4]] – hACT cytosolic + CoA <br /> | ||
**[[4xl4]] – CaACT + CoA <br /> | **[[4xl4]] – CaACT + CoA <br /> | ||
**[[6et9]] – MtACT + PFAM DUF35 + hydroxymethylglutaryl-CoA synthase - ''Methanothermococcus thermolithotrophicus''<br /> | **[[6et9]] – MtACT + PFAM DUF35 + hydroxymethylglutaryl-CoA synthase - ''Methanothermococcus thermolithotrophicus''<br /> | ||
**[[6esq]] – MtACT + CoA + PFAM DUF35 + hydroxymethylglutaryl-CoA synthase <br /> | **[[6esq]] – MtACT + CoA + PFAM DUF35 + hydroxymethylglutaryl-CoA synthase <br /> | ||
**[[6are]] – ACT + CoA + acetate - ''Aspergillus fumigatus'' <br /> | **[[6are]] – ACT + CoA + acetate - ''Aspergillus fumigatus'' <br /> | ||
| + | |||
| + | *SCP2-thiolase | ||
| + | |||
| + | **]]6hrv]] – zfSCP2 - zebrafish <br /> | ||
| + | **]]6hsj]] – zfSCP2 + CoA<br /> | ||
| + | **]]6hsp]] – zfSCP2 + CoA + octanoyl-CoA<br /> | ||
}} | }} | ||
== References == | == References == | ||
<references/> | <references/> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Revision as of 11:01, 20 February 2020
| |||||||||||
3D Structures of Thiolase
Updated on 20-February-2020
References
- ↑ Sundaramoorthy R, Micossi E, Alphey MS, Germain V, Bryce JH, Smith SM, Leonard GA, Hunter WN. The crystal structure of a plant 3-ketoacyl-CoA thiolase reveals the potential for redox control of peroxisomal fatty acid beta-oxidation. J Mol Biol. 2006 Jun 2;359(2):347-57. Epub 2006 Mar 29. PMID:16630629 doi:http://dx.doi.org/10.1016/j.jmb.2006.03.032
- ↑ Soto G, Stritzler M, Lisi C, Alleva K, Pagano ME, Ardila F, Mozzicafreddo M, Cuccioloni M, Angeletti M, Ayub ND. Acetoacetyl-CoA thiolase regulates the mevalonate pathway during abiotic stress adaptation. J Exp Bot. 2011 Nov;62(15):5699-711. doi: 10.1093/jxb/err287. Epub 2011 Sep 9. PMID:21908473 doi:http://dx.doi.org/10.1093/jxb/err287
- ↑ Kiema TR, Harijan RK, Strozyk M, Fukao T, Alexson SE, Wierenga RK. The crystal structure of human mitochondrial 3-ketoacyl-CoA thiolase (T1): insight into the reaction mechanism of its thiolase and thioesterase activities. Acta Crystallogr D Biol Crystallogr. 2014 Dec 1;70(Pt 12):3212-25. doi:, 10.1107/S1399004714023827. Epub 2014 Nov 22. PMID:25478839 doi:http://dx.doi.org/10.1107/S1399004714023827