This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Stathmin
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
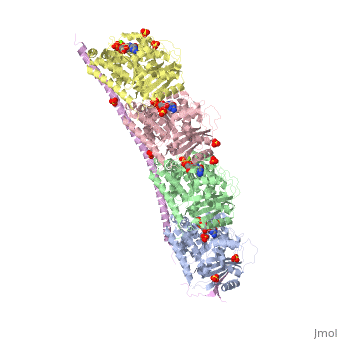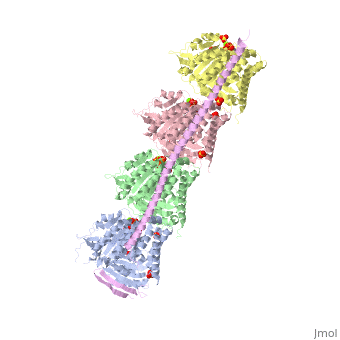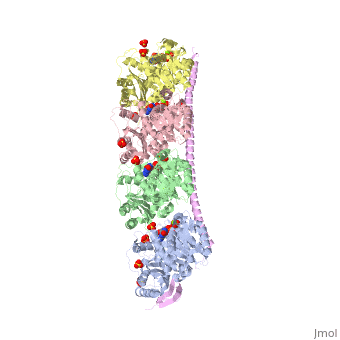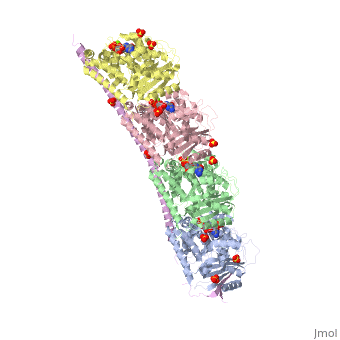
<StructureSection load='3ryf' size='340' side='right' caption='Rat stathmin SLD domain (magenta) complex with tubulin α chain (grey, pink), tubulin β chain (green, yellow), GTP, sulfate and Mg+2 ions (PDB code [[3ryf]])' scene=''> | <StructureSection load='3ryf' size='340' side='right' caption='Rat stathmin SLD domain (magenta) complex with tubulin α chain (grey, pink), tubulin β chain (green, yellow), GTP, sulfate and Mg+2 ions (PDB code [[3ryf]])' scene=''> | ||
== Function == | == Function == | ||
| - | '''Stathmin-4''' (STM-4) regulates microtubules dynamics. Microtubules undergo continuous assembly and disassembly in the cell’s cytoskeleton. STM-4 binds to tubulin and prevents the latter from polymerization thus preventing microtubule assembly. Phosphorylation of STM-4 weakens the binding of STM-4 to tubulin enabling the microtubule assembly needed for the formation of mitotic spindle. Thus, STM-4 is an oncoprotein. STM-4 contains an SLD (Stathmin-Like Domain) domain of 149 residues which binds the tubulin dimer<ref>PMID:15216892</ref>. | + | '''Stathmin-4''' (STM-4) or '''stathmin-like protein B3''' regulates microtubules dynamics. Microtubules undergo continuous assembly and disassembly in the cell’s cytoskeleton. STM-4 binds to tubulin and prevents the latter from polymerization thus preventing microtubule assembly. Phosphorylation of STM-4 weakens the binding of STM-4 to tubulin enabling the microtubule assembly needed for the formation of mitotic spindle. Thus, STM-4 is an oncoprotein. STM-4 contains an SLD (Stathmin-Like Domain) domain of 149 residues which binds the tubulin dimer<ref>PMID:15216892</ref>. |
== Disease == | == Disease == | ||
Current revision
| |||||||||||
References
- ↑ Curmi PA, Gavet O, Charbaut E, Ozon S, Lachkar-Colmerauer S, Manceau V, Siavoshian S, Maucuer A, Sobel A. Stathmin and its phosphoprotein family: general properties, biochemical and functional interaction with tubulin. Cell Struct Funct. 1999 Oct;24(5):345-57. PMID:15216892
- ↑ Cheon MS, Fountoulakis M, Cairns NJ, Dierssen M, Herkner K, Lubec G. Decreased protein levels of stathmin in adult brains with Down syndrome and Alzheimer's disease. J Neural Transm Suppl. 2001;(61):281-8. PMID:11771751