This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Harrison L. Smith/Sandbox1
From Proteopedia
(Difference between revisions)
| Line 14: | Line 14: | ||
The insulin receptor's function in regards to glucose homeostasis is to begin the signaling pathway that will eventually move glucose transporters to the cell surface which will allow glucose to passively defuse into the cell. The glucose receptor is inactive in the absence of insulin. When insulin does bind to the receptor, it undergoes a conformation change, activating it. Once activated, the intracellular Beta subunits autophosphorylate, and downstream signaling begins by the phosphorylation of the Insulin Receptor Substrate (IRS). | The insulin receptor's function in regards to glucose homeostasis is to begin the signaling pathway that will eventually move glucose transporters to the cell surface which will allow glucose to passively defuse into the cell. The glucose receptor is inactive in the absence of insulin. When insulin does bind to the receptor, it undergoes a conformation change, activating it. Once activated, the intracellular Beta subunits autophosphorylate, and downstream signaling begins by the phosphorylation of the Insulin Receptor Substrate (IRS). | ||
===Conformation Change=== | ===Conformation Change=== | ||
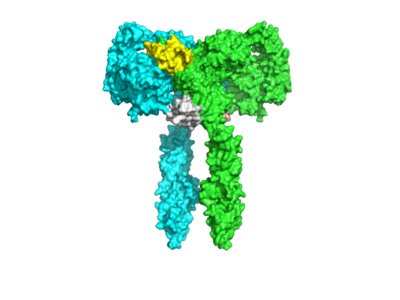
| - | The insulin receptor has two conformations, an active and inactive state. The inactive form predominates in low-levels of circulating insulin, whereas the active conformation is seen when insulin binds to any of the 4 receptor sites. The inactive conformation resembles an inverted V, and the active conformation resembles a T. Upon the binding of insulin to any of the four binding sites, the conformation change will begin, causing the Beta subunit's tyrosine kinase domains to move close together, allowing them to autophosphorylate. This autophosphorylation is what activates the insulin receptor and allows it to participate in further downstream signaling pathways. | + | The insulin receptor has two conformations, an active and inactive state. The inactive form predominates in low-levels of circulating insulin, whereas the active conformation is seen when insulin binds to any of the 4 receptor sites. The inactive conformation resembles an inverted V, and the active conformation resembles a T. Upon the binding of insulin to any of the four binding sites, the conformation change will begin, causing the Beta subunit's tyrosine kinase domains to move close together, allowing them to autophosphorylate. This autophosphorylation is what activates the insulin receptor and allows it to participate in further downstream signaling pathways. <ref name= "Weis"> DOI: https://doi.org/10.1038/s41467-018-06826-6</ref>. |
Revision as of 21:42, 23 March 2020
Homo sapiens Insulin Receptor Ectodomain
| |||||||||||
References
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644
- ↑ doi: https://dx.doi.org/https
- ↑ Wilcox G. Insulin and insulin resistance. Clin Biochem Rev. 2005 May;26(2):19-39. PMID:16278749
- ↑ Riddle MC. Treatment of diabetes with insulin. From art to science. West J Med. 1983 Jun;138(6):838-46. PMID:6351440
Student Contributors
- Harrison Smith
- Alyssa Ritter