Sandbox Reserved 1600
From Proteopedia
(Difference between revisions)
| Line 11: | Line 11: | ||
=Structure= | =Structure= | ||
| - | + | ||
The overall structure contains 19 transmembrane helices that are arranged in a nearly oval shape (Fig 1.) <ref name=”Safarian”>PMID: 27126043 </ref> The protein contains two structurally similar subunits each containing nine helices (blue and red) and one smaller subunit, CydX, with one transmembrane helix. The subunits are interacting using hydrophobic residues and symmetry at the interfaces. The CydX subunit, whose function is not currently known, is positioned in the same way as CydS, which is found in E. coli bd oxidase. Due to its similar structure and position, it has been hypothesized to potentially stabilize heme b558 during potential structural rearrangements of the Q loop upon binding and oxidation of quinol <ref name=”Safarian”>PMID: 27126043 </ref>. The Q loop is shown in lime green, and is a hydrophilic region above Cyd A. The lack of hydrogen bonding in this hydrophobic protein allows the protein to be flexible and go through a large conformational change for reduction of dioxygen. | The overall structure contains 19 transmembrane helices that are arranged in a nearly oval shape (Fig 1.) <ref name=”Safarian”>PMID: 27126043 </ref> The protein contains two structurally similar subunits each containing nine helices (blue and red) and one smaller subunit, CydX, with one transmembrane helix. The subunits are interacting using hydrophobic residues and symmetry at the interfaces. The CydX subunit, whose function is not currently known, is positioned in the same way as CydS, which is found in E. coli bd oxidase. Due to its similar structure and position, it has been hypothesized to potentially stabilize heme b558 during potential structural rearrangements of the Q loop upon binding and oxidation of quinol <ref name=”Safarian”>PMID: 27126043 </ref>. The Q loop is shown in lime green, and is a hydrophilic region above Cyd A. The lack of hydrogen bonding in this hydrophobic protein allows the protein to be flexible and go through a large conformational change for reduction of dioxygen. | ||
==Active Site== | ==Active Site== | ||
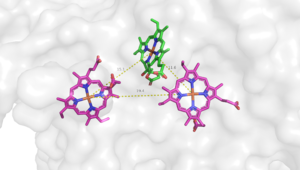
| - | [[Image:Emma 0329 image.png|300 px|right|thumb| | + | [[Image:Emma 0329 image.png|300 px|right|thumb|Figure 1. Heme B558 (pink; left), Heme B595 (pink; right), and Heme D (green)]] |
The active site for Bd Oxidase in ''Geobacillus thermodenitrificans'' is located in subunit Cyd A. The site consists of three iron hemes: Heme B558, Heme B595, and Heme D that are held together in a rigid triangular <scene name='83/838655/Hemes/1'>arrangement</scene> due to van der wals interactions. The length between each heme's central iron is relatively constant which serves to shuttle protons and electrons from one heme to another efficiently. It is suggested that Heme B558 acts as an electron acceptor to the extracellular side and Heme B559 acts as a proton acceptor on the intracellular side. It is then proposed that both heme B558 and B595 shuttle their respective ions directly to Heme D based on this being the shortest pathway (reference). Heme D is then suggested to be the oxygen binding site due to proximity and orientation to the exterior surface of the protein. | The active site for Bd Oxidase in ''Geobacillus thermodenitrificans'' is located in subunit Cyd A. The site consists of three iron hemes: Heme B558, Heme B595, and Heme D that are held together in a rigid triangular <scene name='83/838655/Hemes/1'>arrangement</scene> due to van der wals interactions. The length between each heme's central iron is relatively constant which serves to shuttle protons and electrons from one heme to another efficiently. It is suggested that Heme B558 acts as an electron acceptor to the extracellular side and Heme B559 acts as a proton acceptor on the intracellular side. It is then proposed that both heme B558 and B595 shuttle their respective ions directly to Heme D based on this being the shortest pathway (reference). Heme D is then suggested to be the oxygen binding site due to proximity and orientation to the exterior surface of the protein. | ||
| Line 35: | Line 35: | ||
== Structure Similarity to Bd Oxidase found in ''Ecoli'' == | == Structure Similarity to Bd Oxidase found in ''Ecoli'' == | ||
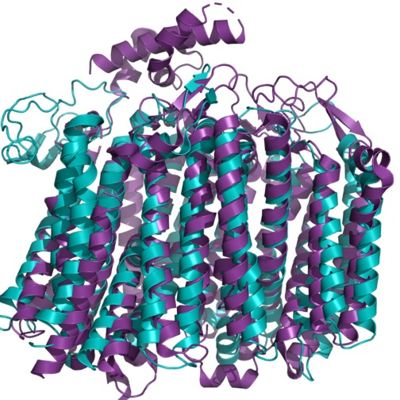
| - | [[Image:Aligmentbdoidase.jpg|400 px|right|thumb|Figure | + | [[Image:Aligmentbdoidase.jpg|400 px|right|thumb|Figure 4. Alignment of bd oxidase for the organisms ''Geobacillus thermodenitrificans'' (blue) and ''Ecoli'' (purple).]] |
Revision as of 23:38, 29 March 2020
bd oxidase; Geobacillus thermodenitrificans
| |||||||||||
References
- ↑ Giuffre A, Borisov VB, Arese M, Sarti P, Forte E. Cytochrome bd oxidase and bacterial tolerance to oxidative and nitrosative stress. Biochim Biophys Acta. 2014 Jul;1837(7):1178-87. doi:, 10.1016/j.bbabio.2014.01.016. Epub 2014 Jan 31. PMID:24486503 doi:http://dx.doi.org/10.1016/j.bbabio.2014.01.016
- ↑ Safarian S, Hahn A, Mills DJ, Radloff M, Eisinger ML, Nikolaev A, Meier-Credo J, Melin F, Miyoshi H, Gennis RB, Sakamoto J, Langer JD, Hellwig P, Kuhlbrandt W, Michel H. Active site rearrangement and structural divergence in prokaryotic respiratory oxidases. Science. 2019 Oct 4;366(6461):100-104. doi: 10.1126/science.aay0967. PMID:31604309 doi:http://dx.doi.org/10.1126/science.aay0967
- ↑ Junemann S. Cytochrome bd terminal oxidase. Biochim Biophys Acta. 1997 Aug 22;1321(2):107-27. doi:, 10.1016/s0005-2728(97)00046-7. PMID:9332500 doi:http://dx.doi.org/10.1016/s0005-2728(97)00046-7
- ↑ Das A, Silaghi-Dumitrescu R, Ljungdahl LG, Kurtz DM Jr. Cytochrome bd oxidase, oxidative stress, and dioxygen tolerance of the strictly anaerobic bacterium Moorella thermoacetica. J Bacteriol. 2005 Mar;187(6):2020-9. doi: 10.1128/JB.187.6.2020-2029.2005. PMID:15743950 doi:http://dx.doi.org/10.1128/JB.187.6.2020-2029.2005
- ↑ Safarian S, Rajendran C, Muller H, Preu J, Langer JD, Ovchinnikov S, Hirose T, Kusumoto T, Sakamoto J, Michel H. Structure of a bd oxidase indicates similar mechanisms for membrane-integrated oxygen reductases. Science. 2016 Apr 29;352(6285):583-6. doi: 10.1126/science.aaf2477. PMID:27126043 doi:http://dx.doi.org/10.1126/science.aaf2477
- ↑ Safarian S, Rajendran C, Muller H, Preu J, Langer JD, Ovchinnikov S, Hirose T, Kusumoto T, Sakamoto J, Michel H. Structure of a bd oxidase indicates similar mechanisms for membrane-integrated oxygen reductases. Science. 2016 Apr 29;352(6285):583-6. doi: 10.1126/science.aaf2477. PMID:27126043 doi:http://dx.doi.org/10.1126/science.aaf2477
- ↑ Safarian S, Rajendran C, Muller H, Preu J, Langer JD, Ovchinnikov S, Hirose T, Kusumoto T, Sakamoto J, Michel H. Structure of a bd oxidase indicates similar mechanisms for membrane-integrated oxygen reductases. Science. 2016 Apr 29;352(6285):583-6. doi: 10.1126/science.aaf2477. PMID:27126043 doi:http://dx.doi.org/10.1126/science.aaf2477
- ↑ Safarian S, Rajendran C, Muller H, Preu J, Langer JD, Ovchinnikov S, Hirose T, Kusumoto T, Sakamoto J, Michel H. Structure of a bd oxidase indicates similar mechanisms for membrane-integrated oxygen reductases. Science. 2016 Apr 29;352(6285):583-6. doi: 10.1126/science.aaf2477. PMID:27126043 doi:http://dx.doi.org/10.1126/science.aaf2477