We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
User:Andre Wu Le Chun/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 13: | Line 13: | ||
| + | [[Image:Spike amino acids.jpg|500px|]] | ||
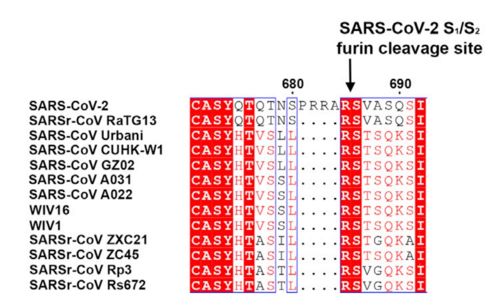
| + | figure 1: Alignment of S in between multiple SARS-CoV (Walls et al., 2020) | ||
== Function == | == Function == | ||
| Line 28: | Line 30: | ||
== Interaction with angiotensin-converting enzime 2 == | == Interaction with angiotensin-converting enzime 2 == | ||
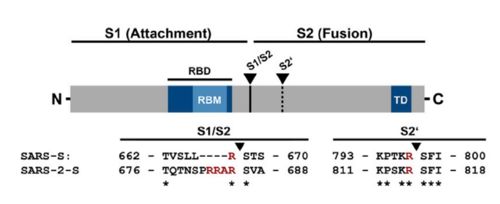
The interaction between the 2019-nCov and the host cell begins with the recognition of the ACE2, a dimeric protein. Then, the S1 subunit moves, modifying the protein's conformation in way that receptor-binding domain becomes available to connect itself to the peptidase domain of target cell. Due to the existence of four amino acids in between the S1 and S2, a furin cleavage site is featured in the protein. The cleavage of this site has potential relevance as it could lead to later priming at the S2' site by a serine protease, TMPRSS2. Once attached on the surface of ACE2, the cleavage of S2' happens and the fusion mechanism is activated. Then, the protein goes through conformational changes, the protein structure assumes a conformation which is suitable for binding with the ACE2 receptor. At that instance, the spike protein is found in a "up" conformation, hence the protein's name. It binds itself to the ACE2 with high affinity (15nM). In order to activate infection, the spike protein is cleaved by TMPRSS2, a host's serine protease, also fundamental for the viral infection. Furine cleavage site | The interaction between the 2019-nCov and the host cell begins with the recognition of the ACE2, a dimeric protein. Then, the S1 subunit moves, modifying the protein's conformation in way that receptor-binding domain becomes available to connect itself to the peptidase domain of target cell. Due to the existence of four amino acids in between the S1 and S2, a furin cleavage site is featured in the protein. The cleavage of this site has potential relevance as it could lead to later priming at the S2' site by a serine protease, TMPRSS2. Once attached on the surface of ACE2, the cleavage of S2' happens and the fusion mechanism is activated. Then, the protein goes through conformational changes, the protein structure assumes a conformation which is suitable for binding with the ACE2 receptor. At that instance, the spike protein is found in a "up" conformation, hence the protein's name. It binds itself to the ACE2 with high affinity (15nM). In order to activate infection, the spike protein is cleaved by TMPRSS2, a host's serine protease, also fundamental for the viral infection. Furine cleavage site | ||
| - | + | [[Image:Spike.jpg|500px|]] | |
| + | figure 2: Alignment between S protein of SARS and SARS-2 (Hoffmann et al., 2020). | ||
</StructureSection> | </StructureSection> | ||
Revision as of 18:43, 21 June 2020
6vsb
Prefusion 2019-nCoV spike glycoprotein with a single receptor-binding domain up
| |||||||||||
References
¹https://www.sciencedirect.com/science/article/pii/S0065352719300284 ²https://www.biorxiv.org/content/10.1101/2020.02.19.956581v1.full
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644