This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
NudT16
From Proteopedia
| Line 5: | Line 5: | ||
<Structure load='6B09' size='350' frame='true' align='right' caption='Crystal structure of HsNUDT16 in complex with diADPR (soaked)' scene='Crystal' /> | <Structure load='6B09' size='350' frame='true' align='right' caption='Crystal structure of HsNUDT16 in complex with diADPR (soaked)' scene='Crystal' /> | ||
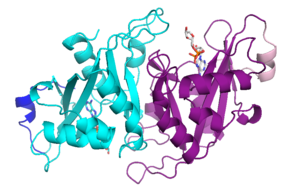
[[Image:Nudt16cartoon.png|300px|right|thumb| NudT16]] | [[Image:Nudt16cartoon.png|300px|right|thumb| NudT16]] | ||
| + | NudT16 is a dimer | ||
Revision as of 15:07, 30 June 2020
Contents |
Introduction
NudT16 is a hydrolase and belongs to the nucleoside diphosphate-linked moiety X (Nudix) family. This protein regulates levels of 53BP1 which is a protein that recruits other proteins to the site of a DNA breakage. NudT16 has also shown in vitro to remove ADP-ribosylation through its hydrolase activities.
Structure
|
NudT16 is a dimer
Function
Disease
Relevance
Structural highlights
</StructureSection>
References
Proteopedia Page Contributors and Editors (what is this?)
Hannah Campbell, Tihitina Y Aytenfisu, Michal Harel, Sandra B. Gabelli