This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
SN2 reaction
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
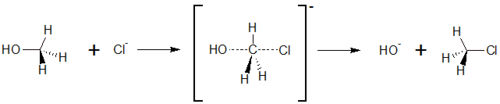
== S<sub>N</sub>2-Substitution of chloride and methanol == | == S<sub>N</sub>2-Substitution of chloride and methanol == | ||
| - | <StructureSection load='' size='340' side='right' caption='' scene='54/542286/Side_view/ | + | <StructureSection load='' size='340' side='right' caption='' scene='54/542286/Side_view/3'> |
SN2 reaction is a basic reaction type in organic chemistry. The letter S<sub>N</sub> stand for nulceophilic Substitution, the number 2 stands for bimolecular. This means that both reactions partners are involved in the reaction rate determining step. It also exists an S<sub>N</sub>1 reaction; here, only one reaction partner is involved in this step. | SN2 reaction is a basic reaction type in organic chemistry. The letter S<sub>N</sub> stand for nulceophilic Substitution, the number 2 stands for bimolecular. This means that both reactions partners are involved in the reaction rate determining step. It also exists an S<sub>N</sub>1 reaction; here, only one reaction partner is involved in this step. | ||
Revision as of 10:29, 8 July 2021
SN2-Substitution of chloride and methanol
| |||||||||||
Proteopedia Page Contributors and Editors (what is this?)
Joel L. Sussman, Jaime Prilusky, Angel Herraez, Verena Pietzner