This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Madison Unger/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 24: | Line 24: | ||
===Tunnels=== | ===Tunnels=== | ||
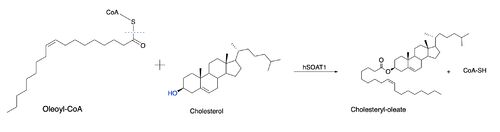
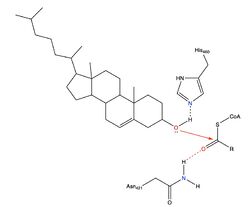
| - | The nine transmembrane segments create a cytosolic tunnel and a transmembrane tunnel that meet at the site of catalysis (active site). Acyl-coenzyme A enters the active site though the cytosolic tunnel and cholesterol enters through the transmembrane tunnel. These then meet in the catalytic site to react and form the cholesteryl ester. When exiting the catalytic site, the free CoA is able to release through the cytosolic tunnel to the cytosol and the cholesterol ester is able to release through the <scene name='87/877505/Active_site_residues/2'>transmembrane tunnel</scene> to the membrane or through the lumen tunnel to the lumen. Certain residues that line the transmembrane tunnel are important in ACAT activity (E259, E263, R262, P304, L306, V423, V424, M265, I261, and H460). | + | The nine transmembrane segments create a cytosolic tunnel and a transmembrane tunnel that meet at the site of catalysis (active site). Acyl-coenzyme A enters the active site though the cytosolic tunnel and cholesterol enters through the transmembrane tunnel. These then meet in the catalytic site to react and form the cholesteryl ester. When exiting the catalytic site, the free CoA is able to release through the <scene name='87/877505/H460_labeled/1'>cytosolic tunnel</scene> to the cytosol and the cholesterol ester is able to release through the <scene name='87/877505/Active_site_residues/2'>transmembrane tunnel</scene> to the membrane or through the lumen tunnel to the lumen. Certain residues that line the transmembrane tunnel are important in ACAT activity (E259, E263, R262, P304, L306, V423, V424, M265, I261, and H460). |
== Medical Relevance == | == Medical Relevance == | ||
The mechanism of ACAT is essential for cholesterol storage and cholesterol transfer through the plasma because cholesteryl ester is the primary form of cholesterol used for these events. Additionally, ACAT can use a variety of different sterol molecules besides cholesterol as substrates and activators. Because of its biological importance, ACAT has been linked to [http://en.wikipedia.org/wiki/Atherosclerosis atherosclerosis], [http://en.wikipedia.org/wiki/Alzheimer%27s_disease Alzheimer’s disease], and cancer as a potential drug target for treatment of these diseases. Various studies have looked into ACAT inhibition and how that inhibition treats or prevents certain diseases, such as reducing the size and metastasis of certain tumors and reducing the formation of plaques in atherosclerosis. ACAT is an important target for these diseases due to its functional relevance in cholesterol metabolism. | The mechanism of ACAT is essential for cholesterol storage and cholesterol transfer through the plasma because cholesteryl ester is the primary form of cholesterol used for these events. Additionally, ACAT can use a variety of different sterol molecules besides cholesterol as substrates and activators. Because of its biological importance, ACAT has been linked to [http://en.wikipedia.org/wiki/Atherosclerosis atherosclerosis], [http://en.wikipedia.org/wiki/Alzheimer%27s_disease Alzheimer’s disease], and cancer as a potential drug target for treatment of these diseases. Various studies have looked into ACAT inhibition and how that inhibition treats or prevents certain diseases, such as reducing the size and metastasis of certain tumors and reducing the formation of plaques in atherosclerosis. ACAT is an important target for these diseases due to its functional relevance in cholesterol metabolism. | ||
Revision as of 20:17, 29 March 2021
ACAT/SOAT
| |||||||||||
References
- ↑ Guan C, Niu Y, Chen SC, Kang Y, Wu JX, Nishi K, Chang CCY, Chang TY, Luo T, Chen L. Structural insights into the inhibition mechanism of human sterol O-acyltransferase 1 by a competitive inhibitor. Nat Commun. 2020 May 18;11(1):2478. doi: 10.1038/s41467-020-16288-4. PMID:32424158 doi:http://dx.doi.org/10.1038/s41467-020-16288-4
- ↑ Qian H, Zhao X, Yan R, Yao X, Gao S, Sun X, Du X, Yang H, Wong CCL, Yan N. Structural basis for catalysis and substrate specificity of human ACAT1. Nature. 2020 May;581(7808):333-338. doi: 10.1038/s41586-020-2290-0. Epub 2020 May, 13. PMID:32433614 doi:http://dx.doi.org/10.1038/s41586-020-2290-0
- ↑ Rogers MA, Liu J, Song BL, Li BL, Chang CC, Chang TY. Acyl-CoA:cholesterol acyltransferases (ACATs/SOATs): Enzymes with multiple sterols as substrates and as activators. J Steroid Biochem Mol Biol. 2015 Jul;151:102-7. doi: 10.1016/j.jsbmb.2014.09.008., Epub 2014 Sep 12. PMID:25218443 doi:http://dx.doi.org/10.1016/j.jsbmb.2014.09.008
Student Contributors
- Leah Goehring
- Gabby Smith
- Anna Campbell