This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Leanne Price/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 27: | Line 27: | ||
===Tunnels=== | ===Tunnels=== | ||
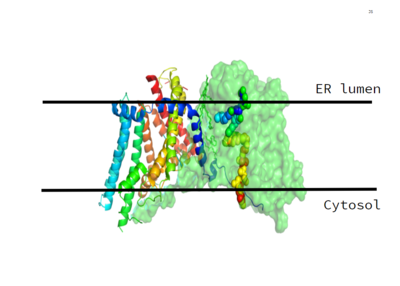
| - | DGAT consists of 3 tunnels, a cytosolic tunnel, an ER-luminal funnel, and a membrane-embedded lateral gate. The cytosolic tunnel is the site of acyl-CoA binding, with the CoA group pointing at the cytosolic face and its acyl chain pointing towards the endoplasmic reticulum lumen. DAG then enters via the lateral gate on the luminal side via the lateral gate where it can then access the active site. The resulting product can then be released to either side of the membrane. <ref name="Sui" | + | DGAT consists of 3 tunnels, a cytosolic tunnel, an ER-luminal funnel, and a membrane-embedded lateral gate. The cytosolic tunnel is the site of acyl-CoA binding, with the CoA group pointing at the cytosolic face and its acyl chain pointing towards the endoplasmic reticulum lumen. DAG then enters via the lateral gate on the luminal side via the lateral gate where it can then access the active site. The resulting product can then be released to either side of the membrane. <ref name="Sui" /ref> |
===Active Site=== | ===Active Site=== | ||
| - | The active site of DGAT is located within the membrane, with the catalytic histidine residue (<scene name='87/877601/His415/5'>His415</scene>-represented in white) buried inside the central cavity. This central cavity serves as the catalytic site. The acyl-acceptor lipid substrates access the active site through the lateral gate within the membrane. The active site also contains <scene name='87/877601/His415_tunnel/2'>His415</scene> and several nearby <scene name='87/877601/His415_and_polar_residues/3'>polar residues</scene> (including Asn378, Gln437, and Gln465) whose side chains are oriented towards the cavity center. These residues interact and create a hydrophilic channel within the active site. The His415 residue is also likely involved in catalysis, making it increasingly significant. In face, single mutations of His415 and Asn378 terminated DGAT activity. This suggests that the central cavity of DGAT within the membrane is the catalytic site. | + | The active site of DGAT is located within the membrane, with the catalytic histidine residue (<scene name='87/877601/His415/5'>His415</scene>-represented in white) buried inside the central cavity. This central cavity serves as the catalytic site. The acyl-acceptor lipid substrates access the active site through the lateral gate within the membrane. The active site also contains <scene name='87/877601/His415_tunnel/2'>His415</scene> and several nearby <scene name='87/877601/His415_and_polar_residues/3'>polar residues</scene> (including Asn378, Gln437, and Gln465) whose side chains are oriented towards the cavity center. These residues interact and create a hydrophilic channel within the active site. The His415 residue is also likely involved in catalysis, making it increasingly significant. In face, single mutations of His415 and Asn378 terminated DGAT activity. This suggests that the central cavity of DGAT within the membrane is the catalytic site. <ref name="Sui" /ref> |
| Line 40: | Line 40: | ||
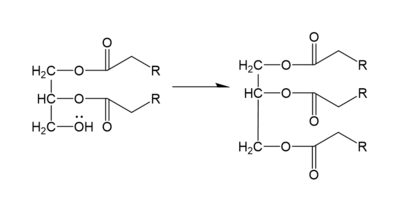
In the DGAT mechanism, the diglyceride serves as the nucleophile. While the acyl group of the CoA enzyme serves as the electrophile. The lone pair on the last hydroxyl group present on the glycerol of the diglyceride attacks the thioester bond of the acyl-CoA enzyme. This attack breaks the sulfur-carbon bond, a weak bond that is easily breakable. This allows the acyl group of the acyl-CoA enzyme to attach to the diglyceride, creating a triglyceride. While the CoA group then serves as the leaving group. | In the DGAT mechanism, the diglyceride serves as the nucleophile. While the acyl group of the CoA enzyme serves as the electrophile. The lone pair on the last hydroxyl group present on the glycerol of the diglyceride attacks the thioester bond of the acyl-CoA enzyme. This attack breaks the sulfur-carbon bond, a weak bond that is easily breakable. This allows the acyl group of the acyl-CoA enzyme to attach to the diglyceride, creating a triglyceride. While the CoA group then serves as the leaving group. | ||
| - | The cleavage site for oleoyl-CoA is within a short distance of a lipid acceptor. This revelation was made after a strong, lipid-like density in the central cavity in the cryo-EM data. Hydrophobic residues line this region and form a channel surrounding the lipid-like density. The channel itself has a bent, hydrophobic pathway that allows the binding of hydrophobic molecules. The bent architecture of this tunnel is likely how DGAT distinguishes acyl acceptors from other molecules, such as cholesterol. | + | The cleavage site for oleoyl-CoA is within a short distance of a lipid acceptor. This revelation was made after a strong, lipid-like density in the central cavity in the cryo-EM data. Hydrophobic residues line this region and form a channel surrounding the lipid-like density. The channel itself has a bent, hydrophobic pathway that allows the binding of hydrophobic molecules. The bent architecture of this tunnel is likely how DGAT distinguishes acyl acceptors from other molecules, such as cholesterol. <ref name="Sui" /ref> |
Revision as of 19:39, 13 April 2021
Diacylglycerol Acyltransferase
| |||||||||||
References
- ↑ 1.0 1.1 Sui X, Wang K, Gluchowski NL, Elliott SD, Liao M, Walther TC, Farese RV Jr. Structure and catalytic mechanism of a human triacylglycerol-synthesis enzyme. Nature. 2020 May;581(7808):323-328. doi: 10.1038/s41586-020-2289-6. Epub 2020 May, 13. PMID:32433611 doi:http://dx.doi.org/10.1038/s41586-020-2289-6
- ↑ Wang L, Qian H, Nian Y, Han Y, Ren Z, Zhang H, Hu L, Prasad BVV, Laganowsky A, Yan N, Zhou M. Structure and mechanism of human diacylglycerol O-acyltransferase 1. Nature. 2020 May;581(7808):329-332. doi: 10.1038/s41586-020-2280-2. Epub 2020 May, 13. PMID:32433610 doi:http://dx.doi.org/10.1038/s41586-020-2280-2
Student Contributors
- Justin Smith
- Eloi Bigirimana
- Leanne Price