This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
AppA protein BLUF domain
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
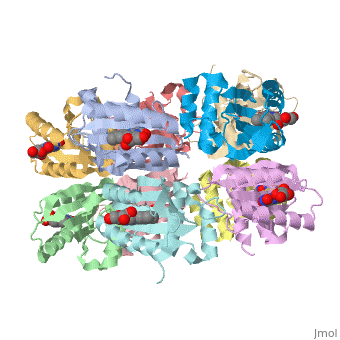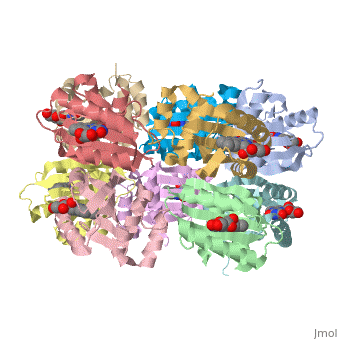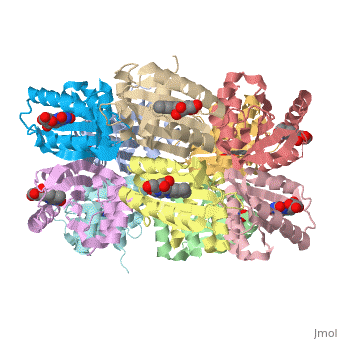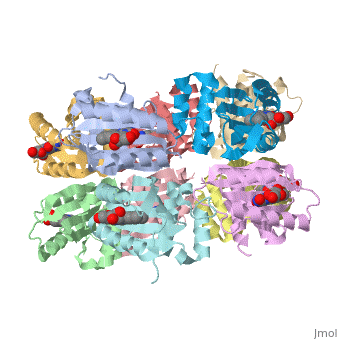
<StructureSection load='1x0p' size='350' side='right' caption='Structure of BLUF domain protein complex with FAD (PDB code [[1x0p]]).' scene=''> | <StructureSection load='1x0p' size='350' side='right' caption='Structure of BLUF domain protein complex with FAD (PDB code [[1x0p]]).' scene=''> | ||
=Introduction= | =Introduction= | ||
| - | '''AppA''' is an anti repressor in which blue light excitation induces a long-lived red-shift absorbance in photosystem synthesis. AppA contains sensor for '''blue light using''' [http://en.wikipedia.org/wiki/Flavin_adenine_dinucleotide FAD] '''(BLUF) domains''' are one class of photoreceptor family that utilizes a flavin [http://en.wikipedia.org/wiki/Chromophore chromophore]<ref name ="one">PMID: 15876364</ref>. The other two classes include [http://en.wikipedia.org/wiki/Phototropin phototropins] (LOV) and [http://en.wikipedia.org/wiki/Cryptochrome cryptochromes]<ref name="two">PMID: 14730990</ref>. The BLUF domain was first discovered in [http://microbewiki.kenyon.edu/index.php/Rhodobacter ''Rhodobacter sphaeroides''] as the blue light photoreceptor involved in the repression of photosynthesis genes in '''AppA''' protein<ref name="one" /><ref name ="three">PMID: 12230978</ref>. The BLUF domain is known to exist in many bacteria, including cyanobacteria. | + | '''AppA''' is an anti repressor in which blue light excitation induces a long-lived red-shift absorbance in photosystem synthesis<ref>PMID:12230978/ref> . AppA contains sensor for '''blue light using''' [http://en.wikipedia.org/wiki/Flavin_adenine_dinucleotide FAD] '''(BLUF) domains''' are one class of photoreceptor family that utilizes a flavin [http://en.wikipedia.org/wiki/Chromophore chromophore]<ref name ="one">PMID: 15876364</ref>. The other two classes include [http://en.wikipedia.org/wiki/Phototropin phototropins] (LOV) and [http://en.wikipedia.org/wiki/Cryptochrome cryptochromes]<ref name="two">PMID: 14730990</ref>. The BLUF domain was first discovered in [http://microbewiki.kenyon.edu/index.php/Rhodobacter ''Rhodobacter sphaeroides''] as the blue light photoreceptor involved in the repression of photosynthesis genes in '''AppA''' protein<ref name="one" /><ref name ="three">PMID: 12230978</ref>. The BLUF domain is known to exist in many bacteria, including cyanobacteria. |
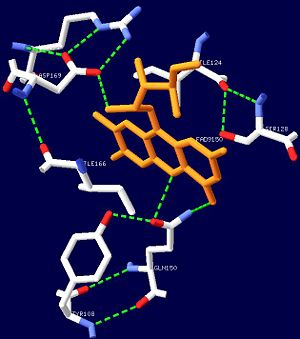
One unique photosensing property of BLUF domain is a light induced spectral shift in the flavin absorption spectrum where the wavelength is longer by approximately 10nm<ref name="one" />. In AppA this shift occurs quickly upon illumination and is slowly reversed upon return to darkness. This spectral shift is not well understood and is not observed in other flavin binding photoreceptors. There have been two proposed models to explain the spectral shift observed in BLUF containing proteins: π-π stacking between the isoalloxazine ring of flavin and the phenol sidechain of a conserved tyrosine residue or protonation and deprotonation of the flavin ring coupled directly or indirectly with the conserved tyrosine residue<ref name ="four">PMID: 14556317</ref><ref name ="five">PMID: 15659451</ref>. These two models are based upon the conserved tyrosine residue Tyr9 in the T110078 protein. | One unique photosensing property of BLUF domain is a light induced spectral shift in the flavin absorption spectrum where the wavelength is longer by approximately 10nm<ref name="one" />. In AppA this shift occurs quickly upon illumination and is slowly reversed upon return to darkness. This spectral shift is not well understood and is not observed in other flavin binding photoreceptors. There have been two proposed models to explain the spectral shift observed in BLUF containing proteins: π-π stacking between the isoalloxazine ring of flavin and the phenol sidechain of a conserved tyrosine residue or protonation and deprotonation of the flavin ring coupled directly or indirectly with the conserved tyrosine residue<ref name ="four">PMID: 14556317</ref><ref name ="five">PMID: 15659451</ref>. These two models are based upon the conserved tyrosine residue Tyr9 in the T110078 protein. | ||
Revision as of 10:30, 12 May 2021
| |||||||||||
3D structures of BLUF domain protein
Updated on 12-May-2021
2byc - RsBlue light receptor BLUF - Rhodobacter sphaeroides
2bun – RsAppA BLUF domain – NMR
2iyg, 2iyi - RsAppA BLUF domain (mutant)
1x0p - BLUF - Thermosynechococcus elongatus
References
- ↑ PMID:12230978/ref> . AppA contains sensor for blue light using FAD (BLUF) domains are one class of photoreceptor family that utilizes a flavin chromophore<ref>PMID: 15876364</li> <li id="cite_note-two-1">[[#cite_ref-two_1-0|↑]] van der Horst MA, Hellingwerf KJ. Photoreceptor proteins, "star actors of modern times": a review of the functional dynamics in the structure of representative members of six different photoreceptor families. Acc Chem Res. 2004 Jan;37(1):13-20. PMID:[http://www.ncbi.nlm.nih.gov/pubmed/14730990 14730990] doi:[http://dx.doi.org/10.1021/ar020219d 10.1021/ar020219d]</li> <li id="cite_note-one">[[#cite_ref-one_16|↑]] <strong class="error">Cite error: Invalid <code><ref></code> tag; no text was provided for refs named <code>one</code></strong></li> <li id="cite_note-three-3">↑ <sup>[[#cite_ref-three_3-0|4.0]]</sup> <sup>[[#cite_ref-three_3-1|4.1]]</sup> Masuda S, Bauer CE. AppA is a blue light photoreceptor that antirepresses photosynthesis gene expression in Rhodobacter sphaeroides. Cell. 2002 Sep 6;110(5):613-23. PMID:[http://www.ncbi.nlm.nih.gov/pubmed/12230978 12230978] </li> <li id="cite_note-four-4">[[#cite_ref-four_4-0|↑]] Laan W, van der Horst MA, van Stokkum IH, Hellingwerf KJ. Initial characterization of the primary photochemistry of AppA, a blue-light-using flavin adenine dinucleotide-domain containing transcriptional antirepressor protein from Rhodobacter sphaeroides: a key role for reversible intramolecular proton transfer from the flavin adenine dinucleotide chromophore to a conserved tyrosine? Photochem Photobiol. 2003 Sep;78(3):290-7. PMID:[http://www.ncbi.nlm.nih.gov/pubmed/14556317 14556317] </li> <li id="cite_note-five-5">[[#cite_ref-five_5-0|↑]] Hasegawa K, Masuda S, Ono TA. Spectroscopic analysis of the dark relaxation process of a photocycle in a sensor of blue light using FAD (BLUF) protein Slr1694 of the cyanobacterium Synechocystis sp. PCC6803. Plant Cell Physiol. 2005 Jan;46(1):136-46. Epub 2005 Jan 19. PMID:[http://www.ncbi.nlm.nih.gov/pubmed/15659451 15659451] doi:[http://dx.doi.org/10.1093/pcp/pci003 10.1093/pcp/pci003]</li> <li id="cite_note-six-6">↑ <sup>[[#cite_ref-six_6-0|7.0]]</sup> <sup>[[#cite_ref-six_6-1|7.1]]</sup> Iseki M, Matsunaga S, Murakami A, Ohno K, Shiga K, Yoshida K, Sugai M, Takahashi T, Hori T, Watanabe M. A blue-light-activated adenylyl cyclase mediates photoavoidance in Euglena gracilis. Nature. 2002 Feb 28;415(6875):1047-51. PMID:[http://www.ncbi.nlm.nih.gov/pubmed/11875575 11875575] doi:[http://dx.doi.org/10.1038/4151047a 10.1038/4151047a]</li> <li id="cite_note-seven-7">[[#cite_ref-seven_7-0|↑]] Yuan H, Anderson S, Masuda S, Dragnea V, Moffat K, Bauer C. Crystal structures of the Synechocystis photoreceptor Slr1694 reveal distinct structural states related to signaling. Biochemistry. 2006 Oct 24;45(42):12687-94. PMID:[http://www.ncbi.nlm.nih.gov/pubmed/17042486 17042486] doi:[http://dx.doi.org/10.1021/bi061435n 10.1021/bi061435n]</li></ol></ref>
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Amanda Cookhouse, Jaime Prilusky