This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1709
From Proteopedia
| Line 21: | Line 21: | ||
<scene name='90/904314/Cap_Domain/1'>VKOR Cap Domain</scene> | <scene name='90/904314/Cap_Domain/1'>VKOR Cap Domain</scene> | ||
Closed conformation | Closed conformation | ||
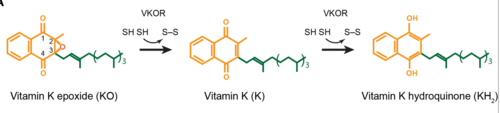
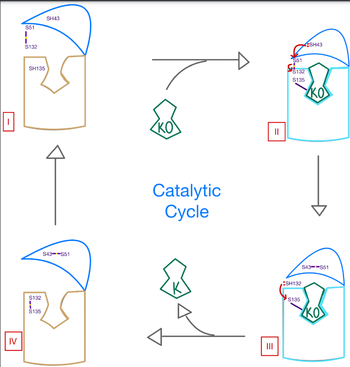
| - | A key part of VKOR is the function of the cap domain | + | A key part of VKOR is the function of the <scene name='90/904314/Cap_Domain/1'>VKOR Cap Domain</scene>, which is located at the top of VKOR towards the intracellular part of the membrane. The cap domain assists with activating Vitamin K as it induces the structural change of VKOR from the open conformation to the closed conformation when the substrate binds. This initiates a domino effect through the [https://reader.elsevier.com/reader/sd/pii/S0021925820001386?token=9F8E1964241D20488CA55E035D35D9A5D650A7B3FDAD9A5579598A8DC00127539BE71CF1785B117102144AC1F41ABB6C&originRegion=us-east-1&originCreation=20220329001707/ catalytic mechanism]. The cap domain has critical interactions that stabilize the closed conformation including a <scene name='90/904314/Disulfide_bridge_stabilization/1'>Disulfide Bridge </scene> between S43 and S51, and polar interactions from D44. |
| - | + | ||
<scene name='90/904314/Disulfide_bridge_stabilization/1'>Disulfide Bridge Stabilization</scene> | <scene name='90/904314/Disulfide_bridge_stabilization/1'>Disulfide Bridge Stabilization</scene> | ||
=== Anchor === | === Anchor === | ||
Revision as of 00:39, 29 March 2022
| |||||||||||
References
1. Li, Weikai et al. “Structure of a bacterial homologue of vitamin K epoxide reductase.” Nature vol. 463,7280 (2010): 507-12. doi:10.1038/nature08720.
2. Liu S, Li S, Shen G, Sukumar N, Krezel AM, Li W. Structural basis of antagonizing the vitamin K catalytic cycle for anticoagulation. Science. 2021 Jan 1;371(6524):eabc5667. doi: 10.1126/science.abc5667. Epub 2020 Nov 5. PMID: 33154105; PMCID: PMC7946407.
3. “Warfarin.” Wikipedia, Wikimedia Foundation, 10 Feb. 2022, https://en.wikipedia.org/wiki/Warfarin.
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644